Chapter Nine
Galley Gear
WHETHER CALLED a kitchen, galley, caboose, chuckwagon, or cookhouse, and whether situated in a home or restaurant, on a ship, freight train, or wagon train, or even outdoors wherever a shelter can be set up, it is a place dedicated to the vital task of preparing food for anyone from a sole diner to an army. Its barest essentials are a few pots of clay or metal, sources of heat and water, and perhaps a knife. All else is excess.
And in today’s kitchens, boy, do we have excess!
We have refrigerators; gas, electric, and induction ranges; convection and microwave ovens; mixers; blenders; nonstick cookware; and—well, just look around your kitchen, you lucky dog. You’ve come a long way, baby.
But just as these tools have to be in proper shape to deal with a variety of foods, we have to know how to deal with the tools themselves. There is nothing more frustrating to a craftsman than having to repair a tool before being able to use it.
Does your dishwasher eat your aluminum utensils? Does your refrigerator exude an uninvited fragrance? Does your butter keeper spoil your butter? Does your oven cook a roast faster or slower than the recipe says? Do your pizzas come out flabby and your cakes goopy?
It’s all in how you use your armamentarium of appliances, apparatuses, equipment, tackle, gear, gadgets, and utensils. Treat them with understanding and respect, for as Emerson wrote, “If you do not use the tools, they use you.”
Or, to paraphrase Thoreau, you become the tools of your tools.
THE FOODIE’S FICTIONARY: Microwave—a baby’s bye-bye gesture
IN SEARCH OF SMELLICULES
I’ve always kept an open box of baking soda in my refrigerator to absorb odors. But I’ve noticed that there’s now a new kind of baking soda box in the supermarket that supposedly works even better, even though the label says it contains nothing but pure baking soda. How does baking soda absorb odors, and how does this new contraption do it better?
Like every other householder in this country, I have religiously kept an open box of Arm & Hammer (is there any other kind?) baking soda in my refrigerator, and I can testify that I have never smelled a bad odor. It must also have worked to repel tigers, because not once did I encounter a tiger in my house as long as that baking soda box was in the fridge.
Is it possible that I never saw a tiger because I live so far from India, or that I never smelled a foul odor in my refrigerator because I’m such a fastidious fridgemeister? Nah! Not according to the Arm & Hammer Division of Church & Dwight Co., Inc., and every domestic maven in the U.S.A., who would staunchly maintain that the baking soda absorbed all the odors. (They make no claims about tigers.)
What hard evidence do we have that baking soda really works, at least for odors? None that I know of. But here’s the theory.
Baking soda is pure sodium bicarbonate(NaHCO3), also known as bicarbonate of soda. It reacts with both acids and bases, that is, with both acidic and alkaline chemicals. (The bicarbonate ion is amphoteric.) But it is more than twenty times as effective in reacting with acids as with bases. And thereby hangs the odor-eating theory. Should a wandering molecule of a smelly acid alight upon a surface of baking soda, it will be neutralized, turned into a salt (shades of Lot’s wife!), and trapped permanently. True enough. There is no arguing with the fact that baking soda will gobble up acids—if given the opportunity. But there’s the rub, or rubs. How do we get the acid to come into contact with the baking soda, and why do we want to trap acids anyway?
First, why are acids the alleged stinkers? It goes back mostly to spoiled milk. In the old days of undependable refrigeration, and especially before pasteurization, milk quickly spoiled, not only by bacterial growth but by its butterfat breaking down into fatty acids, primarily butyric, caproic, and caprylic acids. Butyric acid is largely responsible for the odor of rancid butter, whereas caproic and caprylic acids are named after what they smell like: caper is Latin for goat. Get the drift?
So if you are in the habit of leaving month-old milk in the refrigerator for several weeks while you visit your time-share, many of the sour fatty acid molecules may indeed find their way to an open box of baking soda, fall in, and be neutralized.
But not all smelly molecules (smellicules?) that can pollute your refrigerator’s air space are acids, or even bases (alkalis) for that matter; chemically speaking, they can be virtually anything. Claiming that baking soda absorbs “odors” generically is stretching the truth by a chemical mile.
Let’s put it this way: An odor is a puff of gaseous molecules floating through the air to our noses. Each type of molecule has its unique chemical identity and its own unique set of reactions with other chemicals. No single chemical, sodium bicarbonate included, can claim to react with and deactivate any and all gaseous chemicals that happen to smell bad.
Even for acidic odors that are bicarbonate’s main quarry, note that the landing pad for a smellicule on a box of baking soda is a mere 7 square inches (the box-top area) located at some random position within a 20-cubic-foot (35,000-cubic-inch) refrigerator air space. That’s not a very efficient system for capturing smellicules. The box does not attract odors, as many people believe. It has no come-hither power, in spite of its toplessness.
The new “contraption” you saw is Arm & Hammer’s creatively spelled Fridge-n-Freezer Flo-Thru Freshener, a standard one-pound box of baking soda with removable sides, intended to give gaseous molecules more access to the baking soda by “flo-ing thru” porous paper inner seals. That sounds like a great idea, but this box, “specially designed to expose more baking soda than any other package,” uncovers only another 7 square inches of baking soda surface. And the air in the fridge doesn’t “flo thru” the package anyway. There is no fan or other force blowing it into one side of the box and out the other. Nice advertising concept, though.
In short, as it says on Arm & Hammer’s website, “Arm & Hammer Baking Soda’s deodorization power is legendary!”
I agree. It’s a legend.
What about the odor molecules that baking soda won’t absorb? There is only one common substance that can gobble them all up indiscriminately: activated charcoal. It works not by trying to be a chemical for all seasons, but by using a physical stickiness that is essentially chemistry-blind. If gases find their way into its enormous interior network of microscopic pores, they stick by a phenomenon called adsorption, the adhesion of molecules to a surface by means of what chemists call van der Waals forces.
Charcoal is made by heating hardwood, nutshells, coconut husks, animal bones, or other carbon-containing materials in an oxygen-free environment, so that they don’t actually burn, while substances other than carbon are driven off. It is then “activated” by being treated with very high temperature steam, a process that removes any remaining noncarbon substances and results in an extremely porous microstructure within the charcoal grains. A single gram (one twenty-eighth of an ounce) of activated charcoal may contain up to 2,000 square meters (18,000 square feet) of internal surface area.
You may be able to find activated charcoal (the best kind is made from coconut husks) in a drugstore, hardware store, appliance store, or pet shop. Spread it out on a baking pan with sides, and leave it in the offending fridge for a couple of days. Do not use charcoal briquettes; they contain coal, sawdust, and other substances, and their charcoal content wouldn’t work anyway because it isn’t powdered or activated.
In the end, there is only one sure-fire route to a sweet, odor-free refrigerator. Three words: prevention, prevention, prevention. Seal all your refrigerated food, especially “strong” foods such as onions, in airtight containers. Check frequently for signs of spoilage, round up the usual suspects, and throw them away. Wipe up spills promptly. Clean the fridge thoroughly. Yeah, I know, but do it more often.
Oh, you say there was a power outage while you were on vacation and all your refrigerated food spoiled and you could smell it all the way from the airport on your return? Poor soul. Neither baking soda nor charcoal will help, nor will cursing the power company. Make yourself a stiff drink, go to Louisiana State University’s disaster information website http://www.lsuagcenter.com/Communications/pdfs_bak/pub2527Q.pdf, and follow the directions.
BUTTER KEEPERS DON’T
Sticks of butter stored in a covered glass butter dish in the butter-keeper compartment in the door of our refrigerator develop a dark yellow, slightly rancid-tasting skin. Is there any way to prevent this?
You probably think you’re doing everything right, don’t you? Well, the worst place to keep butter is in a butter dish, and the worst place to keep the butter dish is in the “butter keeper” of your refrigerator.
Butter dishes were invented to facilitate serving, not preserving. Because they’re not airtight, the butter’s surface is exposed to air and can oxidize and become rancid.
Butter compartments should be banned. Many of them have little heaters inside to keep the butter at a slightly warmer temperature than the rest of the fridge to make it easier to spread. But the warmer temperature speeds up oxidation of the fat.
I keep my butter in the freezer compartment, tightly enveloped in plastic wrap. Yes, it’s hard as a rock when I want to use it, but a sharp knife can whack off a piece that will warm up and soften rather quickly.
SAINTS (AND CHEMICALS) PRESERVE US!
I’ve always wondered why some foods go bad so quickly even if refrigerated, while others seem to last forever without refrigeration. Things like opened mustard and ketchup bottles can last for weeks outside the refrigerator, and cheese, honey, and peanut butter can survive at room temperature for even longer. Is there any general way to estimate how long a food will last?
Would that life were that simple! There can be no single rule that covers all the foods we consume—an almost infinite number of combinations of thousands of different proteins, carbohydrates, fats, and minerals that make up our omnivorous diet. “Going bad” can refer to the effects of bacteria, molds, and yeasts; heat; oxidation from exposure to air; or enzymes in the foods themselves. Enzymes in fruits, for example, are there specifically to hasten their ripening, maturing, and ultimate decay.
One thing is inevitable, however: all foods will eventually spoil, rot, decompose, disintegrate, crumble, putrefy, turn rancid, or become just plain yucky. It’s Nature’s law, for dust they art and unto dust shalt they return. Proteins will turn soft, squishy, putrid, and green; carbohydrates will ferment and sour; fats will turn rancid. Ketchup and mustard keep so long because they contain microbe-inhibiting acid (vinegar), no fat, and no active enzymes.
In battling food spoilage, we humans have cooked our foods, smoked them, dried them, acidified them, and salted or sugared them—and, thanks largely to an American inventor named Clarence Birdseye (yes, that was really his name), in recent decades we have frozen them.
During a stint as a fur trader in Labrador, Birdseye watched the native people freeze fish and meats for later consumption. He noted that when frozen quickly in the winter instead of more slowly during the milder times, the food retained better texture, flavor, and color when thawed.
In 1925, Birdseye unveiled his “Quick Freeze Machine,” and the frozen-food industry was off and running. Today, Birds Eye Foods bills itself as the nation’s largest processor of frozen vegetables. And no, there never was a Mr. Jolly Greengiant.
Freezing preserves foods because the frozen water, a.k.a. ice, is unavailable for use by spoilage microorganisms, so they can’t grow. Refrigeration, as distinguished from freezing, will slow their growth, but there are limits. At a typical home-refrigerator temperature, ten thousand bacteria can become ten billion in a few days.
Enter preservatives: chemicals added to prepared foods to extend their shelf lives—and the lives of us who eat them. Yes, preservatives are chemicals. And yes, they are also additives, because, obviously, they have been added. (So have salt, sugar, spices, vitamins, and so on.) Quite simply, without preservatives most of our foods would spoil. And yet we are continually wooed by food labels demurely hinting at their superiority with the phrase “Contains no additives or preservatives.” Someday I’d like to see a label that adds “Will spoil almost as soon as you get it home.”
What are these chemicals? They fall mostly into four categories.
- Antimicrobials inhibit the growth of bacteria, molds, and yeasts. They include the sulfur dioxide and sulfites used in fruits, fruit juices, vinegars, and wines; sorbic acid used in cheeses; calcium propionate and other propionates used to inhibit molds in bread and other baked goods; and sodium and other benzoates used to prevent fungal growth in beverages, fruit preserves, cheeses, pickles, and many other products. Benzoates occur naturally in cranberries, while propionates can be found in strawberries, apples, and cheeses.
- Antioxidants inhibit oxidation by air, which makes fats, especially unsaturated fats, turn rancid. They include sulfites (again), BHA (butylated hydroxyanisole), BHT (butylated hydroxytoluene), TBHQ (tertiary butylated hydroquinone), ascorbic acid (vitamin C), and propyl gallate. They’re used in potato chips, nuts, cereals, and crackers.
- Enzyme inhibitors slow spoilage by enzyme-driven reactions in foods. Sulfites (again) inhibit enzymatic degradation reactions in fruits such as raisins and dried apricots. Acids, such as ascorbic acid and the citric acid in lemon juice, deactivate enzymes, including the enzyme phenolase, which makes apples and potatoes start turning brown as soon as they are cut.
- Sequestrants, also known as chelating agents, gobble up atoms of trace metals such as iron and copper that catalyze (accelerate) oxidation reactions and cause discoloration. The most widely used chelating agent is EDTA or ethylenediamine tetraacetic acid (pronounced ETH-ill-een-DYE-a-meen-…oh, never mind). Other sequestrants are polyphosphates and citric acid.
Okay, so some of these chemical names are hard to pronounce. But contrary to the opinions of some, that doesn’t make them evil. They’re all added in tiny amounts regulated by the FDA, and nobody eats them by the spoonful.
Your alternative to eating foods containing preservatives is to visit the farm or farmers’ market every day for fresh meat and produce. Also, make your own cream, preserves, pickles, cheese, wine, potato chips, cereals, and olive oil, being sure to consume them before they go bad.
And welcome to the eighteenth century.
NO NUKES
When we travel abroad and return to the United States, we’re not allowed to bring home plants or foods for public health reasons. But aren’t these items sterilized when they pass through the airport security X-ray machines?
No. Airport security X-rays are not nearly intense enough to kill insects, parasites, and the like. The radiations used to sterilize and preserve foods are millions of times as intense.
BASICALLY, IT’S BASIC
I wonder why aluminum cookware and utensils become discolored and seemingly corroded in my dishwasher. An aluminum mesh strainer went particularly fast. Is this because there is an acid condition in the soap or water?
No, it’s not an acid. It’s the chemical opposite of an acid: an alkali, known to chemists more accurately as a base.
Most dishwasher detergents for machines, as opposed to the dishwashing detergents for hand-washing dishes, contain the highly alkaline compound sodium carbonate, also known as good old-fashioned washing soda—not baking soda, which is sodium bicarbonate.
Alkaline chemicals are needed in the dishwasher because they gobble up grease, transforming it into soap. A soap is one of a class of chemical compounds formed by the action of an alkali on a fat. A detergent, on the other hand, is a more modern synthetic compound specifically designed to do soap’s cleaning chores. That difference doesn’t stop many people from calling all of today’s household detergents “soaps” anyway.
But I digress.
We tend to think that if a chemical is attacking and dissolving a metal, it must be an acid. And that’s generally true; a strong enough acid could devour a Humvee and spit out the tires. But aluminum is an unusual metal in that it is attacked by both acids and alkalis. (It is amphoteric.) So the alkaline sodium carbonate in the dishwasher detergent does indeed attack aluminum, at the very least eating deeply enough into the surface to make it dull and pewter-gray with aluminum compounds. For this reason most manufacturers of quality aluminum cookware advise against putting it in the dishwasher.
Worse yet, some dishwasher detergents contain potassium hydroxide or sodium hydroxide (lye), which are much stronger alkalis than sodium carbonate and will literally eat into your aluminum utensils. That’s probably what converted your mesh strainer into a basketball hoop. If you still want to wash your aluminum cookware in the dishwasher, scan the labels of the dishwasher detergents in your supermarket and choose one that contains neither potassium or sodium hydroxide nor sodium carbonate. They do exist.
There’s a second aluminum-damaging phenomenon going on in your dishwasher if the aluminum utensil happens to be touching another metal, which will most likely be stainless steel. Whenever any two different metals, in this case aluminum and what is essentially iron, are in contact while immersed in an electrically conducting liquid, an electrical (more properly, an electrolytic) reaction takes place that attacks one of the two metals, in this case the aluminum, corroding its surface and dulling it. So if you insist on washing an aluminum utensil in the dishwasher, make sure it isn’t touching any other kind of metal.
Sidebar Science: Stop, thief!
FOR REASONS that are more elaborate than we want to get into here, iron atoms hold on to their electrons more tightly than aluminum atoms do. (Iron is said to be more electronegative than aluminum.) Thus, if the two metals happen to be in contact within an electrically conducting liquid (an electrolyte) such as dishwasher detergent dissolved in water, the iron atoms will actually steal electrons from the aluminum atoms. This transfer of electrons constitutes a flow of electric current, with the electrolyte completing the circuit.
The now-electron-deficient aluminum atoms (aluminum cations) want to regain their normal complement of electrons by reacting with something—anything—that has electrons to donate. The surface of the aluminum metal therefore reacts with negative ions (anions) in the solution, forming a dulling layer of an aluminum compound, most often aluminum oxide.
THE EMULSION COMPULSION
I’m confused about emulsions. Some recipes tell me I’m emulsifying certain ingredients, but all I can see is that I’m merely blending them. Is there some special trick I’m missing?
No, but I feel your pain. The word emulsify is too often misused as a synonym for blend or thicken. It is not. Restaurant menus love to call any thick sauce an emulsion. It is not. Chefs like to say they’re emulsifying a sauce by using a roux. They’re not.
Many substances, including flour, cornstarch, gelatin, pectin, okra, egg, and even pureed banana, will thicken a soup, custard, jam, gravy, or sauce. But when you use them you are not emulsifying anything. An emulsion is a very specific kind of mixture of two liquids that don’t ordinarily mix, one suspended in the form of tiny droplets within the other.
The prototypical kitchen example of a true emulsion is mayonnaise, in which the mutual loathing between oil and water (the latter existing within the vinegar or lemon juice and egg white) has been overcome by two things: the brute force of being beaten together, and the action of a special chemical ingredient called an emulsifier. Only when this combination of physical and chemical powers is operating will oil and water mix and stay mixed in the form of a true emulsion.
As on that blind date we’ve all suffered through at one time or another, there are simply no attractive forces between a water molecule and an oil molecule. So even if you shake a bottle of vegetable oil and vinegar until they appear to have coalesced into a homogeneous whole, they will sooner or later, usually sooner, separate into two distinct layers. You will have failed to make a stable emulsion.
At most, you will have made a colloidal suspension, in which the oil has been broken down into such tiny microdroplets or globules that they are kept suspended in the vinegar by the constant bombardment of water molecules from all directions. But this marriage is doomed to failure. No matter how much muscle power you put into shaking your vinaigrette dressing, even with the assistance of a governor of California, the oil globules will eventually bump into one another and reunite into a coherent, separate layer. Again, no permanent emulsion.
We can foil the reunification of the oil globules by adding a secret ingredient called an emulsifying agent or an emulsifier. Emulsifiers are made of snakelike molecules that have long, oil-loving (lipophilic) tails and water-loving (hydrophilic) heads. Their lipophilic tails burrow into the oil globules, leaving their hydrophilic heads sticking out like thousands of cloves studding a ham. The “cloves” attract a cloak of water molecules because they contain positive and negative charges that pull on the water molecules’ slightly negative and positive parts. (Water molecules are dipoles.) The resulting cloak of water molecules disguises the oil droplet as a water lover and prevents other oil droplets from attempting to unite with it. Because the emulsifier cloaks all the oil droplets in this way, they will not coalesce even if they bump into one another. They remain individually suspended. Now we have a true emulsion.
Where can we find those secret agents called emulsifiers? An excellent one is lecithin, a phosphorus-containing, fatlike chemical (a phospholipid) found in egg yolks. The phosphorus ends of its molecules are hydrophilic and their other ends are lipophilic. In mayonnaise, they emulsify the oil and vinegar into a permanently stable, homogeneous sauce.
Because we make mayonnaise from a small amount of vinegar or lemon juice (water) and a large amount—about eight times as much—of oil, it may be hard to believe that all that oil has been crowded into that small amount of water. Many people are thus led to believe that mayonnaise is a suspension of tiny water droplets in oil, rather than a suspension of tiny oil droplets in water. But in fact there are so many oil globules that they aren’t actually suspended in the vinegar so much as coated with a very thin film of water, as in a bucket of wet peas, which tend to stick together because of the water’s surface tension. That’s why mayonnaise is so thick.
How emulsifier molecules make oil and water compatible. The zigzag, fat-loving (lipophilic) tails of the emulsifier molecules penetrate the oil globule, leaving their water-loving (hydrophilic) “eyes” sticking out, thereby giving the globule a water-loving surface.
It is true, however, that a suspension of tiny water droplets in oil—the opposite of mayonnaise—is also classified as an emulsion. Butter and margarine, for example, are emulsions of water in oil.
When making mayonnaise with a whisk, we must dribble the oil into the acid-and-egg mixture very slowly to ensure that every added bit of oil is promptly reduced to colloidal-sized globules. If the oil drops are too big to remain suspended, they will coalesce while floating up to the top, where they will form a separate layer, defeating the entire purpose. After a while the oil can be added a little faster, because relatively large droplets will be quickly surrounded by millions of already emulsifier-coated colloidal globules that will “insulate” them from one another, keeping them apart temporarily until they themselves get whacked down to colloidal size.
When making mayonnaise in a blender, on the other hand, we can add a small amount of oil directly to the vinegar-and-egg mixture just before turning on the machine. The blender blades are much quicker than a whisk at chopping the oil globules down to colloidal size—so quick that the globules don’t have time to coalesce.
Commercial dressings and other foods contain a wide variety of emulsifiers to keep their complex mixtures of carbohydrates, fats, proteins, and water together in a stable form. Some of the emulsifiers that you may see on ingredient labels are mono-and diglycerides, polyglycerol esters, propylene glycol esters, and sugar esters of fatty acids. And, of course, lecithin.
Foods are often thickened by substances such as gelatin, starches, and gums, including agar, acacia, xanthin, and carrageenan. But thickeners aren’t emulsifiers. Thickeners work by making the watery part so viscous that even relatively large oil droplets can’t rise through it to coalesce into a layer.
Any old trick to keep a food homogeneous. Who wants to eat oily puddles mixed with globs of watery paste?
THE FOODIE’S FICTIONARY: Hollandaise—Dutch Week at Epcot Center
Sidebar Science: Mixing it up
IN COOKING, we are continually mixing and blending ingredients. But there are several distinct kinds of mixtures. Emulsions are only one of them.
A combination of solid particles, such as salted and peppered flour or a blend of dried spices, is a simple physical mixture. But when liquids are involved, a mixture can take on any of several forms.
• Solution: The most homogeneous mixture of all is a solution, in which the individual molecules or ions (electrically charged atoms or groups of atoms) of one substance are dispersed intimately, molecule beside molecule, among those of the other. Examples are alcohol or sugar dissolved in water, where the alcohol or sugar molecules are intimately mixed in among the water molecules cheek by jowl—if they had cheeks and jowls. Another example is the tomato’s red coloring compound, lycopene, when dissolved in an oil. (Notice how the fat in your tomato-containing recipes always turns red? The color is dissolved lycopene.)
• Colloidal suspension: Many other food mixtures are colloids, or colloidal suspensions, in which invisibly small but generally bigger-than-a-molecule particles of one substance (millionths to thousandths of an inch in size) are suspended throughout the other substance, which is most often a liquid. The particles are held in suspension against the pull of gravity because they are continually being bombarded from all sides by the molecules of the substance in which they are dispersed. The liquids within plant and animal cells are colloidal protein particles suspended in water-based solutions.
• Emulsion: An emulsion is similar to a colloidal suspension. In an emulsion, formed by the action of an emulsifying agent, slightly-larger-than-colloid-sized globules of one liquid are suspended in another liquid with which it wouldn’t ordinarily mix. Mayonnaise and hollandaise sauce are the best-known examples in the kitchen.
Smoky Garlic Mayonnaise
Smoked Spanish paprika, pimentón, adds a subtle hint of wood fire to this garlic mayonnaise. It is a classic accompaniment to paella (chapter 6), and is also good with Hot-Wok Mussels (chapter 6). You can use it as a dip for raw vegetables, or serve it with steamed fish, especially with fresh cod. If the flavor is too intense, half olive oil and half peanut oil makes a delicious dressing.
For herb mayonnaise, add ½ cup minced fresh herbs (parsley, chives, chervil, tarragon) in place of the garlic in step 2. A blender does the best job of blending the herbs into the emulsion.
1 large egg
1 teaspoon smoked Spanish paprika (pimentón)
½ teaspoon dry mustard
½ teaspoon salt
2 tablespoons cider, sherry, or wine vinegar
1 cup mild extra-virgin olive oil
1 large clove garlic, coarsely chopped
1. Break the egg into the blender container. Add the pimentón , mustard, salt, and vinegar. Add ¼ cup of the oil. Cover the container and turn the motor on to low speed.
2. Immediately uncover and pour in the remaining oil in a fine, steady stream. Do not hurry. When all the oil has been incorporated, add the garlic (or the herbs, if using). Continue to blend for 1 minute, or until smooth.
3. Allow the mayonnaise to rest in the refrigerator for at least 1 hour before using, so the flavors will mellow and soften. Refrigerate for up to 4 days. Don’t serve it cold, because chilling dulls the olive oil flavor.
MAKES ABOUT 1¼ CUPS
HOME ON THE RANGE
Oven temperatures are pretty easy to control; the dials have actual temperature numbers on them. But what about stovetops? I have a gas cooktop, and the controls are marked “hi,” “med,” and “lo.” Two of them burn at higher Btu’s than the other two; “med” on them is hotter than “hi” on the other two. I used to have an electric cooktop with the same markings, but their cooking speeds were completely different from my gas range. Are there any industry standards for burner temperatures?
Unfortunately not. The only standard that I know of seems to be that high is spelled “hi” and low is spelled “lo.” In between “hi” and “lo,” my gas range has the digits 2 through 9, but the numbers indicate nothing whatsoever about temperature. The labels “hi” and “lo” and the numbers 2 to 9 refer not to the temperature but the rate at which the burner is generating heat.
There is a lot of confusion about the words heat and temperature in the food world, so maybe it’s “hi time” for me to give you the “lodown.”
First of all, heat and temperature are two different things. Heat is a form of energy, distinct from gravitational energy, electrical energy, energy of motion (kinetic energy), or nuclear energy. It is, in fact, the ultimate form of energy into which all other forms eventually degenerate. (See “The energy tax,” chapter 9.)
Cooking employs heat to cause physical and chemical changes that we hope will improve the food’s tenderness, digestibility, and flavor. It should come as no surprise that when a food (or anything else) absorbs heat, it gets “hotter,” meaning that its temperature rises. But what is temperature? It’s nothing more than a convenient number invented by humans (Mssrs. Fahrenheit and Celsius; see “Untangling F & C,” chapter 9) to indicate how much heat energy a substance contains. In cooking, specific changes take place when a food reaches specific temperatures, that is, when the food acquires enough heat relative to its size. You might say that temperature measures the concentration of heat in a substance.
So it’s the temperature of the food, not the temperature of the gas flame or electric burner beneath the pot or pan, that matters to the cook. The burner is there only to pump heat into the food, no matter what its own temperature may be while doing it. We could place a white-hot poker beneath a frying pan, but it would be a terribly inefficient way to heat the food in the pan.
Then why do we say that one burner at a given setting is “hotter” than another? It’s just loose talk; we don’t really mean to imply that its temperature is higher. We mean only that that burner pumps out heat at a faster rate than the other one, thereby raising the food’s temperature—and cooking it—faster. Instead of “hi” and “lo,” then, we should really label the burner settings “fast” and “slow” (or, inevitably, “slo”).
Different burners, whether gas or electric, do indeed pump out heat at different rates. We measure those rates in Btu’s per hour. A Btu, or British thermal unit, is an amount of heat energy, just as a calorie is. (A nutritional calorie happens to be almost exactly equal to four Btu’s.) But what’s important about a cooktop burner is how many Btu’s or calories it can pump out per minute or per hour. The number of Btu’s pumped out per hour is a good indication of how fast a burner will heat and cook our food. A candle, for example, gives off a total of about 5,000 Btu’s of heat over a period of a few hours, but that’s hardly fast enough to cook with, because its Btu-per-hour rate is pathetic.
Most people, including appliance salesmen and cookbook authors, either are too lazy to say “Btu’s per hour” or don’t know the difference, so they (as you did in your question) speak simply of “Btu’s” as if they were a measure of heating speed. But as Tony Soprano would say, wha’y’gon’do?
A home gas or electric range burner may put out between 9,000 and 15,000 Btu’s per hour at their maximum settings. Check the literature that came with your range or contact its manufacturer to find out the ratings of your burners, and you’ll know which ones are hotter (whoops! I mean faster).
In cooking, what ultimately counts is how fast the temperature of the food rises to its optimum cooking temperature and how steadily it will remain there at different burner settings. But alas, the burner’s setting can be only a rough guide, because no matter what its Btu-per-hour rating, most of the heat it generates goes into heating up the kitchen.
Experience with a given cooktop will teach you approximately what each combination of burner and setting can accomplish. But good cooks simply keep an eye on what the food itself is doing, continually judging whether more or less heat is called for and adjusting the burner accordingly.
Life is tough.
Untangling F & C
In 1714 a German glassblower and amateur physicist named Gabriel Fahrenheit (1686–1736) made a gadget that would indicate how hot or cold an object was by how far up or down a thin column of mercury inside a glass tube would expand or contract as its temperature changed. To put numbers on it, he decided that there should be 180 “degrees” between the freezing point and the boiling point of water. Then he made up a batch of the coldest concoction he could create—a mixture of ice and ammonium chloride—and called its temperature zero. When he stuck his gadget into freezing water, the mercury went up 32 degrees higher than that. Since boiling water was to be 180 degrees higher than that, it came out to be 212. And that’s how we got those crazy numbers, 32 and 212.
Six years after Fahrenheit himself cooled to room temperature, a Swedish astronomer named Anders Celsius (1701–1744) decided that it would be more convenient if there were only 100 “degrees” between the freezing and boiling points of water. So he set the freezing point at zero and the boiling point at 100. And that’s how we got the Celsius scale of temperatures.
Every chance I get, I lobby for a little-known, simple method of conversion between Fahrenheit and Celsius temperatures. (And yes, it’s in all my other books, and I’ll keep doing it until everybody gets it right!) Forget those confusing formulas you learned in school. (Do you add—or is it subtract—32 before—or is it after—multiplying—or is it dividing—by 5/9? Or is it 9/5?)
My way is as easy as 1-2-3:
(1) Add 40 to the number you want to convert (either F or C).
(2) Multiply or divide the result by 1.8.
(3) Subtract 40.
That’s it. All you have to remember is the fact that Fahrenheit temperatures are always bigger numbers than their Celsius equivalents, so you multiply by 1.8 to convert from C to F, and you divide by 1.8 to convert from F to C.
Example: 212°F + 40 = 252
252 ÷ 1.8 = 140
140 – 40 = 100°C
…and that’s just what you expected it to be, right?
Sidebar Science: The energy tax
HEAT, THE energy we use in cooking, is the most universal form of energy. All other energy forms—chemical energy, energy of motion (kinetic energy), electrical energy, nuclear energy—eventually degenerate into heat, which you might say is a sort of energy of last resort. As chemical reactions give off energy, as moving things slow down, as a lamp converts electrical energy into light, as uranium atoms convert mass into radioactivity and heat, there can never be a 100 percent conversion. Inefficiency seems to be built into the universe. Some of the lost or converted energy must inevitably be “wasted” by being turned into heat. You might think of heat as a tax on the conversion of energy; it’s like the fee charged by a money exchanger for converting one form of currency into another.
Most forms of energy can be well-behaved. For example, kinetic energy is well-behaved when a truck is moving in a straight line down a highway. Electrical energy is well-behaved when the movement of electrons is being controlled by a circuit. Nuclear energy comes from the very carefully controlled splitting of atoms. In contrast to all this, however, heat is a shockingly ill-behaved and disorderly form of energy, because it consists of the wild, random movement of atoms and molecules.
The science of thermodynamics has found that whenever a form of energy, such as the chemical energy in a truck’s diesel fuel or in the uranium of a nuclear reactor, is being converted into another form, the disorderliness or randomness (the entropy) of the system must increase. That’s the Second Law of Thermodynamics. The universe is inexorably winding down by losing energy and creating disorder. Chaos.
So whenever a form of energy is being used up or converted into another, some of a disorderly, more chaotic, higher-entropy form of energy must be produced. That’s heat.
For further details, consult your friendly neighborhood thermodynamicist.
FOILING BROILING
I’ve been trying to get some answers to questions about oven broilers. They seem to be the most inconsistent of all kitchen equipment. I’ve moved several times and have had several broilers. One may sear a steak beautifully, while another steams it before it can get brown. How far from the heat should the food be? What about gas versus electric? Preheat or not? Door open or closed?
You’re right to be befuddled. Of the six basic methods of cooking, broiling is the hardest to control.
What are the six basic methods, you ask? They are (1) immersion in hot water or stock (boiling, poaching, stewing); (2) exposure to hot water vapor (steaming); (3) immersion in hot oil (deep-frying); (4) contact with hot metal (pan-frying, sautéing, searing, grilling); (5) exposure to hot air (baking, oven roasting); and (6) exposure to infrared radiation. That last-named method is what we call broiling. (Okay, add the absorption of microwaves to the list if you wish.)
Maybe you think you don’t broil with infrared radiation. But the molecules of anything that’s hot, such as the flame or heating element in a broiler, are emitting infrared radiation, a kind of electromagnetic energy that other molecules can absorb with the result that they become hot in turn. You can feel the warming of the molecules in your face when you walk by anything that’s hot, such as a red-hot furnace or even a range burner that you forgot to turn off. So any cooking method that involves a source of heat—and what method doesn’t? (okay, except microwaves)—is cooking the food at least partially by shooting infrared radiation at it.
Broiling cooks food almost entirely by infrared radiation. The heat source, whether a red-hot electric element or a line of gas flames, doesn’t touch the food; it bathes it in intense infrared radiation, which is absorbed by the top surface of the food, heating it to 600 to 700°F (320 to 370°C) and searing and browning it quickly. Then, after you turn the food over, the same thing happens on the other side.
In electric ovens set on “broil,” only the top heating element gets hot, and the food is placed close beneath it. In some gas ovens, the burner may be beneath the oven floor, doing double duty by also heating the oven, so the food to be broiled must be placed even below that, usually in a drawer-like arrangement.
But we all learned in school that heat rises, didn’t we? So how come we can cook food beneath the source of heat? Well, pardon me, but heat doesn’t rise. Heat from a hot object can flow up, down, or sideways. It will flow into any cooler object with which it happens to be in contact. What people mean when they say heat rises is that hot air rises. Heated air expands and becomes less dense, so it floats upward through the denser, cooler air like a bubble in water.
And while we’re picking nits, what we usually call grilling—that is, placing food above red-hot charcoal or gas flames—can also be called broiling because it’s not the rising hot air that cooks the food so much as the infrared radiation. Even so, largely because of the charcoal’s smoke and the juices that drip down onto hot surfaces and vaporize, this kind of broiling imparts very different flavors to the food than “indoor” broiling does.
Broiling is a good cooking method for tender meats, poultry, and fish, because it’s a dry, high-temperature, short-time method. Less tender meats generally need long, moist cooking to break down the collagen in their connective tissue. Beef steaks and other red meats are a natural for broiling, while pork, chicken, and fish have to be watched carefully to prevent their drying out.
The biggest question in broiling is how close the meat should be to the heating element or gas flames, because a small difference in distance can make a big difference in temperature. The right distance will depend on the type and thickness of the meat, on its fat content, and especially on the idiosyncrasies of the broiler itself. As you’ve noticed, your broiler isn’t your mother’s or your neighbor’s broiler. They’re all different. In general, though, the top surface of the meat should be 3 to 6 inches from the heat source, thin meat relatively closer and thick meat farther away so it can cook through before its surfaces char.
Should you leave the door open? Usually it’s left open in electric ovens to prevent hot-air baking and to let the smoke out. In stove-bottom gas broilers, the drawer is kept closed because the flames consume the smoke, and leaving it open could make a greasy mess on your kitchen floor.
Should you preheat the oven? It’s generally not necessary, although I’ve seen an almost equal number of “always preheat” and “never preheat” admonitions. The best advice—in fact the only good advice—is to follow carefully the directions in the broiling chart in the instruction manual that came with your oven. The manufacturers have spent a lot of time and effort to determine the best conditions for broiling various kinds of meat in their equipment. If you’re one of those people who throw away instruction manuals, or if you can’t find yours (have you looked in your kitchen’s “everything else” drawer?), you can usually order a free replacement from the manufacturer.
Remember that the fat-catching broiler pan that came with your oven is an important part of the picture, so don’t expect to get the same results with any old pan of perhaps a different size. When I use the recommended broiler pan, shelf height, lack of preheating, door ajar, and cooking time for broiling chicken in my electric oven, it comes out perfectly, even though it looks to me as if the chicken is much too close to the heating unit and the door is open too wide. It doesn’t pay to second-guess manufacturers. They know their stuff best.
TWO TIMES ONE EQUALS 1.8
The instructions for my microwave oven tell me how long to reheat one serving of this or that. But sometimes I want to reheat two or more servings at the same time. To reheat x servings, should I set the timer for x times the number of minutes recommended for a single serving?
No. Heating two servings of something takes less than twice the amount of time required for heating one.
Different foods absorb microwaves to different degrees. Water and fats absorb microwaves efficiently, while proteins and carbohydrates don’t absorb much at all. That’s why different foods require different amounts of time to heat or cook. Furthermore, the microwave generator (the magnetron) varies its output according to how big a “load” of absorbing material (a.k.a. food) is in the oven.
Here’s a very rough way of looking at the problem. Let’s say that your particular food absorbs—and turns into heat—a certain percentage of the magnetron’s microwave output. But when there are two servings in the oven, neither one is being exposed to the magnetron’s full output of microwaves; each gets only the un-absorbed “leftovers” from the other. So naturally it will take more time to heat two of them than to heat one. But how much more?
I’ll spare you the arithmetic, but the way it works out is that if one of your servings absorbs, say, 40 percent of the microwaves that it is exposed to, then it will take only 25 percent more time to heat two portions than to heat one. This time increase won’t always be 25 percent; it will be different for different foods that have different appetites for absorbing microwaves.
I tested these ideas with my own “smart” microwave oven, which has pre-programmed cycles for various common heating and cooking chores. For “Heating a Beverage,” for example, the oven first asks me to press a button to tell it how much liquid I want to heat. It then begins its pre-programmed heating cycle for that amount. I timed the heating periods, and here’s how long they lasted: for 0.5 cup, 30 seconds; for 1.0 cup, 50 seconds; for 1.5 cups, 70 seconds; for 2.0 cups, 90 seconds. You can see that the first half-cup requires 30 seconds but that each additional half-cup requires only 20 additional seconds. Two cups took only 1.8 times as much time (90 ÷ 50) as a single cup.
Another example: For “Baked Potatoes” (they’re actually not being baked, but I’ll let that go), the oven cooks one potato in 4½ minutes, and adds 3 minutes and 10 seconds for each additional potato. Putting it another way, two potatoes take only 1.7 times as long as a single potato; three potatoes take 2.4 times as long, and four potatoes take 3.1 times as long.
Lacking an omniscient oven that has pre-programmed cycles for every conceivable type and amount of food, all we can do is make an educated guess. For two servings, your first guess should be about one and three-quarters times the time required for a single serving. Doubling the time would be likely to overheat your food, perhaps making it splatter or dry out. It’s best to be conservative, because you can always zap it a little longer.
Sidebar Science: Say what?
MICROWAVE OVENS are very complicated devices, truly understood only by their electrical-engineer designers. The analysis above, based on a constant percentage of microwave energy being absorbed by each portion, is oversimplified. But I didn’t think you wanted to get involved in load impedances, cavity resonances, and loss constants. And neither did I, for the plain reason that I don’t understand them.
NOTHING BEATS-A PIZZ-A
A new pizza restaurant opened recently in my area, with everything imported from Italy, from the furniture to the brick oven to Roberto, the owner, who actually earned a diploma in pizza-making in Naples. He attributes much of the superb quality of his pizzas to the brick oven. Also, some home cooks I know swear by their pizza stones for baking pizzas and crusty breads. Is there really something to these claims, and if so, what’s so special about brick and stone?
It’s true. Dough, whether for pizza or for bread, baked on a stone surface such as the floor of a brick oven or one of those flat stoneware oven accessories called pizza stones, really does come out crisper and browner than if it were baked on a metal baking sheet or pan. If the oven walls are also stone or brick, so much the better. Early bakers had to build their ovens out of available natural materials such as stone and bricks made of clay. Today we bake our bread in “improved,” technologically sophisticated ovens made of steel. And ironically, they don’t do nearly as good a job.
Brick and stone have two properties that make them work so well: high heat capacity and high emissivity.
Heat capacity is a technical term meaning, well, the capacity to hold heat. If a substance has a high heat capacity, it can absorb a lot of heat without its temperature going up very much. That resistance to having its temperature changed cuts both ways: during heating and during cooling. Once the substance has had its temperature raised, it doesn’t want to cool down any more than it wanted to heat up, so it retains its temperature for a relatively long time.
Stone and brick have higher heat capacities than metals. For the same thickness, an oven floor made of fire clay has twice the heat capacity of iron and two and a half times the heat capacity of copper. So once heated to the desired temperature (and that may take a long time), a clay floor holds its heat well, staying uniformly at that temperature and resisting temperature changes, such as when relatively cold dough is placed on it. Note also that the larger the mass of a material, the higher its capacity to hold heat, just as a bigger pitcher can hold more water. That’s why massive brick ovens with thick floors and walls have always been valued for their baking prowess. On a smaller scale, that’s also why a heavy frying pan “holds its heat” (that is, stays at a constant temperature) better than a thin one.
Brick, clay, and stone have a second, even more powerful advantage over metallic oven materials: their vastly superior emissivities.
Infrared (loosely called “heat”) radiation in a hot oven is absorbed by the molecules of the materials it strikes, which then re-emit much of the radiation almost instantly. In some substances, notably metals, most of the absorbed radiation is dissipated before it can be re-emitted. Only a fraction of the absorbed radiation (16 percent in the case of a stainless-steel oven wall) is returned promptly to its environment: the air in the oven. (In techie talk, the emissivity of a stainless-steel surface is 0.16.) The rest of its heat stays in the oven wall and is wasted, as far as the food is concerned, except that it can slowly and inefficiently work its way back into the air.
Even at the same temperature, then, stone emits more infrared radiation than metal does. And because infrared radiation doesn’t penetrate beyond the surfaces of materials, more infrared radiation striking the dough results in better browning and crisping of its surface.
So whether you’re reheating a delivered pizza, making one from scratch, or baking a free-form loaf of bread, place it on a preheated pizza stone. If the stone is unglazed and therefore porous, it will have the additional advantage of absorbing the steam emitted from the bottom surface of the dough, keeping it dry for even more effective crisping.
Sidebar Science: Heat capacity and emissivity
• Heat Capacity: Let’s take water as the most familiar example of a material that has a relatively high heat capacity.
When we heat water, we’re pumping calories of heat into it; its temperature will therefore rise. Temperature is a measure of how fast the molecules are moving. Because water molecules stick quite tenaciously to one another (by dipole-dipole attraction and hydrogen bonding), it’s relatively difficult to goose them into moving faster. We have to add a whole (nutritional) calorie of heat in order to raise the temperature of a kilogram (a liter) of water by a single degree Celsius. (That is, the specific heat of water is one kilocalorie per kilogram per degree C.) Conversely, when water cools, it has to lose a lot of heat—that same one nutritional calorie per kilogram—for its temperature to be reduced by a single Celsius degree.
A couple of consequences of these facts are that (1) it takes “forever” for a heated pot of water to come to a boil, and (2) a body of water, such as a large lake or an ocean, moderates the surrounding climate by refusing to heat up or cool down as easily as the land does.
• Emissivity: In any environment above absolute zero in temperature—and that includes all environments—there is infrared radiation flying through the space. When such radiation strikes a surface, the molecules in that surface absorb some of it. They exhibit the fact that they now contain more energy by moving more agitatedly: twisting, rotating, and tumbling like a hyperactive kindergarten class during a Ritalin shortage. Each kind of molecule has its own unique ways of rotating and tumbling, corresponding to the unique, characteristic sets of energies that it is capable of absorbing. (That is, different molecules have different infrared absorption spectra.)
After absorbing the radiant energy, the excited molecules “calm down” by re-emitting some of it. Some kinds of molecules re-emit virtually all the energy they had absorbed, while others retain some, converting it into different forms of energy. A substance that re-emits 100 percent of the energy it absorbs is said to have an emissivity of 1.00. (In Techspeak, it behaves like a black-body radiator .)
In general, metals have very low emissivities because their loose electrons can soak up the energy like a sponge. Aluminum, for example, re-emits only 5 percent of the infrared radiation that strikes it; copper, only 2 percent. In contrast, materials such as stone and brick re-emit virtually all of the radiation they absorb: 90 percent for dark brick, 93 percent for marble, 97 percent for tile; that is, their emissivities are 0.90, 0.93, and 0.97, respectively. That’s because the molecules in these substances are fixed rigidly in place, and can’t retain the energy by oscillating and tumbling. In these materials, very little infrared energy is wasted; almost all of the infrared radiation that strikes these stonelike surfaces is re-emitted toward the food.
BAKING BY TOOTHPICK
Why do the directions on cake-mix boxes tell us to lower the oven temperature by 25°F if we’re using a glass cake pan or dish instead of a metal one?
Not all of the cake-mix boxes tell us that. In a perusal of the acres of cake-mix boxes on the shelves of my supermarket (in space consumption probably second only to breakfast cereals), I found, as expected, a wide variety of baking instructions, specifying a wide variety of baking times and temperatures for different pan sizes, shapes, and materials. And that’s not even considering the plight of those unfortunates who live at high altitudes, who are exhorted to modify almost everything from the time and temperature to the amounts of flour and water.
The necessity of changing the time and temperature for various pan shapes and sizes is easy to explain. It’s a matter of surface-to-volume ratio. That is, if the same volume of batter is spread out into a wide pan, exposing a large surface area to the oven’s heat (a large ratio of surface to volume), such as in a sheet cake, it will cook faster than if it were poured into a bundt pan, which exposes relatively little surface area to the hot air.
Then there’s the question of what the pan is made of. In my supermarket survey I found that for standard, shiny aluminum pans, almost all the mixes specify a preheated oven temperature of 350°F (177°C). For dark-colored pans, many of the boxes specify a lower temperature of 325°F (163°C). Several boxes specify 325°F for glass baking pans, but several also say 325°F for glass or metal pans, without mentioning dark-colored pans at all. And one devil-may-care box, bless its heart, says “350°F (any type pan).”
So what’s a guy to do?
I am now going to violate the most fundamental principle of expository writing, if not of teaching, by admitting at the start that none of the recommendations matter in the end, and then asking you to bear with me while I explain the scientific reasons behind the recommendations.
- The color of the pan: A relatively shiny aluminum or stainless-steel cake pan obviously reflects visible light more than a dark-colored anodized one or a nonstick-coated one. Because all the light falling on an object must be either reflected or absorbed, that means that the dark surface is absorbing more light than the shiny one is. That extra absorbed light energy makes the dark-colored pan slightly warmer than the shiny one, even in a same-temperature oven. (To manage our body heat, we wear lighter-colored clothes in the summer and darker ones in the winter.)
But what light is there inside a dark oven, you ask? Infrared radiation, which many people refer to as infrared “light” even though it’s invisible to the human eye. A dark surface absorbs more of this radiation than a light-colored or shiny surface does. That’s particularly important because when an object absorbs infrared radiation it becomes warmer—significantly warmer than if it had absorbed visible light. Thus, a cake should cook faster in a dark pan than in a light one, and we are often advised to lower the oven temperature to compensate.
- The material of which the pan is made: A thin metal cake pan of any color conducts the oven’s heat efficiently into the batter. But compared with metal, a glass pan is a very poor conductor of heat and is quite sluggish at transmitting its oven-given heat into its contents. Using a glass pan and given the choice of fast baking at a high temperature or slower baking at a lower temperature, we would choose the latter, because the oven’s heat needs a longer time to penetrate through the glass to the batter. It’s not a big effect, so a relatively small decrease in oven temperature or a small increase in baking time (which some cake-mix instructions specify) suffices.
And now, as promised, I repeat that none of this really matters. Home ovens don’t work like the carefully calibrated equipment in the test kitchens of Betty Crocker or Duncan Hines. There, armies of lab-coated technicians painstakingly work out the best possible conditions for baking their mixes to ensure that the home cook garners accolades from his or her family and runs back to the store to purchase more. But in real life, home ovens may vary from their set temperatures by plus or minus 25°F (14°C) or even more, and the issue of 350 versus 325 degrees is in most cases moot.
So use whatever kind of pan you have, and by all means, turn your oven dial to the recommended number. But don’t bet your cookies on it. After all the directives about pan material, oven temperature, and baking time, every cake-mix box that I’ve seen winds up admitting that the cake is done when, and only when, it looks done and a toothpick inserted into it comes out clean.
And that’s the truth.
Sidebar Science: How ovens cook
WE SPEAK of oven temperature as the main variable that determines how fast a cake will bake or any food will cook. But although the temperature is of primary importance, it is only one factor. Even at exactly the same oven temperature, the amount of heat energy a food actually receives and absorbs may not be the same.
By “oven temperature,” we mean the temperature of the air inside the enclosure, and that’s what the temperature control device regulates. But once the air is heated to a certain temperature, there are still three ways in which the air’s heat can be transmitted into the food: by conduction, by convection, and by radiation.
• Conduction: When two substances at different temperatures are in contact, such as hot oven air in contact with a food’s surface, heat will flow from the higher-temperature air into the lower-temperature food by the process of conduction. Just as water always flows downhill if it can, heat will always try to flow “down-temperature” from high to low. The heat energy is conducted from the air to the food across their interface by direct molecular collisions. That is, the hot air molecules are moving faster than the cooler food molecules (that’s actually the definition of temperature: it’s the average motion energy—kinetic energy—of the molecules), and when they collide with the food’s molecules they kick them up to a faster (hotter) speed, like a cue ball scattering a rack of billiard balls.
But conduction is very inefficient. Air molecules are separated from one another by interplanetary distances, relatively speaking, so the chances that a hot air molecule will collide with the surface of a cake pan or a roast are small. Heat conduction can be quite efficient between two solids in contact, such as your hand on a hot frying-pan handle, but not between hot air and anything else. You can put your hand in a 200°F (93°C) oven for several seconds without fear, because the rate of conduction of heat from the air into your skin is so extraordinarily slow. But don’t try dipping your hand into 200°F water. Water is a much better conductor of heat because its molecules are much closer together than air’s are.
And why are metals the best heat conductors of all?
In almost all other materials, the atomic electrons are parts of individual molecules. But in metals, the electrons belong, in effect, to all the atoms simultaneously. We can think of metal atoms as being embedded in a swarm or sea of shared electrons, like raisins in an electron pudding. When a metal comes in contact with the agitated molecules of a hot substance, it’s the electron swarm that transfers the agitation—the heat—rapidly to all other parts of the metal. That’s heat conduction.
In an oven, however, the other two heat transmission mechanisms, convection and radiation, are more important than conduction.
• Convection: Variable conditions inside the oven, such as inevitably uneven temperatures between one spot and another, make the air move, because hotter “pieces” of air rise, while cooler “pieces” fall, creating a kind of circulation that’s called convection, or convection currents. This circulation boosts the efficiency of heat transfer between the air and the food, because it increases the amount of contact between the food and the hot air molecules in the enclosure. A convection oven enhances this effect by means of a fan that circulates the oven’s internal air or some externally heated and blown-in hot air, leading to more efficient heat transfer and faster cooking. That’s why it’s good practice to lower the temperature by about 25°F (14°C) when using a convection oven rather than a standard one.
• Radiation: The third mechanism by which food becomes hot in an oven is by absorbing radiation. The oven’s heating element or flame and its walls and floor are hot—they are what make the air hot—and hot things radiate infrared radiation. In fact, all materials at all temperatures are emitting some of their energy as infrared radiation. (See “Heat capacity and emissivity” on chapter 9.)
For a given object, the hotter it is, the more infrared radiation it is emitting. When the infrared radiation coming from the hot oven walls and the hot air hits the food, the food molecules absorb it and move with increased energy. That is, they become hotter.
Infrared radiation isn’t heat, as many books will tell you. It is electromagnetic radiation, like radio, radar, and microwaves, but of just the right wavelength to be absorbed by most kinds of molecules, which thereby become more energetic and hotter. I call infrared radiation “heat in transit,” because it is emitted by hot matter and travels through space, yet it isn’t transformed back into heat until it is absorbed by other matter.
OVEN RUBBER
More and more, I’m seeing kitchen gadgets such as spatulas and pastry brushes made of silicone. What amazes me is the baking pans and muffin “tins,” which look and feel like rubber, but can supposedly stand oven temperatures up to 500°F. What’s the secret?
The secret, as Julius Caesar might have said, is that all rubber is divided into three parts. Or, in somewhat more modern language, that which we call rubber by any other name would not bake as well.
I’ll try again. There are three basic kinds of rubber, coming from three different kinds of plants: natural rubber, which comes from latex, the sap of the tropical tree Hevea brasiliensis; synthetic rubber, which is made in a chemical plant; and silicone rubber, which comes from, well, a different chemical plant. The last two were dreamed up by chemists in attempts to duplicate some of natural rubber’s unique properties and improve upon others.
A synthetic rubber called neoprene was first marketed by DuPont in 1931, and a wide variety of silicone rubbers have been manufactured by General Electric and Dow Corning since the 1940s. These two man-made products inherited the silly name rubber from the natural material, which was so christened by the English chemist and clergyman Joseph Priestley in 1770, when he found that it would rub out pencil marks, if not erase sins.
Unfortunately, in recent times the word silicone has been implanted, so to speak, in the public’s mind in but a single context: cosmetic augmentation. But silicones are a remarkably versatile family of chemical compounds with hundreds of uses. In culinary applications, the French fiberglass-reinforced silicone baking-pan liner called Silpat has been used in professional kitchens since it was introduced in 1982. But silicones have only recently invaded the American home kitchen in many forms, all approved by the FDA for repeated contact with food. Today, the whole baking pan, not just its liner, is made of silicone.
Before I go any further, I must straighten out some terminology, because the words silicone and silicon are so often mistakenly interchanged.
Silicon (no e) is a chemical element, the second most abundant element on Earth (after oxygen). It is a rock-hard, brittle gray material that would make the world’s worst cake pans, not to mention surgical implants. However, silicon the element is a semiconductor, and therefore immensely valuable in the form of “chips” or microprocessors in computers and hundreds of other electronic devices. That’s why the high-tech region around San Jose, California, is called Silicon Valley. (It is to be carefully distinguished from Los Angeles, which has been dubbed Silicone Valley for reasons I need not explain.)
Silicones, on the other hand, are chemical compounds that, like the natural and synthetic rubbers, are polymers, meaning that their molecules consist of long chains of thousands of smaller molecules tied together. Silicone molecules have spines made of alternating atoms of silicon and oxygen, to which are attached various groups of carbon and hydrogen atoms. Depending on the lengths of the chains and the identities of the attached groups, silicones can range from liquids (used in brake fluids and water-repellent sprays) to gels (in breast implants) to greases (in lubricants and lipsticks) to elastomers, the rubber-like materials in Silly Putty, Superballs, refrigerator door gaskets, and, now, kitchenware.
Silicone bakeware has a remarkably useful set of properties. First of all, the material is inherently translucent, so a veritable kaleidoscope of bright colors can be incorporated into the products. (Kitchen-Aid’s line of muffin pans, loaf pans, and cake pans comes in red or blue.) Silicone pans can withstand high temperatures without melting (without their molecules flowing apart from one another) because the molecules are very long and tightly intertwined, like a plate of cold, leftover Spaghetti with Glue Sauce. That’s also why you can take them directly from the oven to the freezer or vice versa without any fear of cracking; the molecules, while individually flexible, are so rigidly fixed in place that the material can’t expand or contract very much with changes in temperature.
Silicones don’t absorb microwaves, but like all microwave-safe utensils they can get hot in the microwave oven from contact with the heated food. Because silicones are chemically inert, the pans are dishwasher-safe; caustic detergents can’t touch them. Also because of their nonreactivity, they are more or less nonstick; cakes and muffins release easily—most of the time—since you can flex the pans to pop them out. But don’t try to use them as Jell-O or aspic molds; sitting the mold in a warm water bath won’t release the gelatin because the silicone is a heat insulator.
Any disadvantages? Being electrical insulators (one of the most important properties of silicone rubbers in other applications), they are subject to static electricity and may collect dust in the pantry between uses. And their floppiness can be disquieting, for example when you’re carrying a batter-filled pan to the oven. Carry the pan on a rimless baking sheet, using the sheet as a peel when inserting the pan into the oven.
Caveat emptor department: As with everything else, there are high and low qualities of silicone bakeware. Remember that “silicone” isn’t a single chemical material. Dow Corning, for example, sells dozens of different silicone formulations with different properties, for fabricators to use in molding their commercial products. Some may not be as heat-resistant as others, so check the maximum temperature ratings on the labels. They can range from 450°F (232°C) up to 675°F (357°C) for silicone trivet pads.
SHAPE MATTERS
Recipes are always telling us to roast something at a certain temperature for a certain length of time. But then they tell us to test it near the end to see whether it’s done, and in my experience it almost never is. Shouldn’t the recipe developer be able to give me a more exact cooking time?
The quick and dirty answer is no; there are just too many uncontrolled variables.
The cruel fact of life is that when a recipe tells you to cook for “x hours at y degrees,” it’s only a guideline, an educated guess, a ballpark estimate. It’s what worked—most of the time—for the elves who tested the recipe, but there’s no guarantee it will work for you. So, sorry, Virginia, but there is no Santa Claus. (I’ve been wanting to straighten that kid out for years.)
Except perhaps in a food research laboratory, there is no such thing as a standard roast on a standard rack in a standard pan at a standard position in a standard oven at a carefully regulated oven temperature. Each one of these factors can vary, producing different results even if all other things were equal. But as Wolke’s Law of Pervasive Perversity says, “All other things are never equal.”
You can’t just go around saying that a beef or pork roast or a chicken or turkey should be cooked for so many minutes per pound at a certain temperature. Even if Wolke’s Law didn’t apply and you could magically control everything else, the one variable that you have no control over is the most important one: the shape of the roast. Not its weight but its shape: how much surface area it presents to the oven’s heat. Heat can enter the meat only through its surface, so the more surface area a roast has for its weight, the faster it will cook.
Here’s an example.
If we had two roasts of the same weight—that is, the same volume—one shaped like a cube and the other shaped like a sphere, the cubic roast would have 24 percent more surface area than the spherical one. That’s just geometry. Work it out yourself if you get your kicks that way. For my part,
I never saw a cubic cow
I never hope to see one.
But I can tell you anyhow,
It’ll roast about 24 percent faster than a spherical one.
Another example: Suppose we cut that cubic roast in half parallel to one face. Its surface area will then be increased by 33 percent. The two halves, then, should cook in roughly 33 percent less time than the whole one.
So again, dear, naïve little Virginia, no Santa Claus, or even a reasonably good fairy, exists who can weigh your irregularly shaped rib roasts or turkeys and tell you exactly how many minutes per pound to cook it, even if Wolke’s Law were repealed.
TIME AND TEMPERATURE WAIT FOR NO HAM
I want to roast a piece of meat in an oven for 24 hours at 180°F (82°C). Would this use less gas or electric energy than roasting it for 3 hours at 375°F (191°C)? How about 6 hours at 250°F (121°C)?
This may sound like an odd question, but it was asked of me by the food authority and author Paula Wolfert when she was working on her book The Slow Mediterranean Kitchen: Recipes for the Passionate Cook. Her concept was that long, slow cooking can produce tender, juicy, flavorful meats that higher-temperature cooking cannot match. And as usual, she’s right, as the recipes in her book amply demonstrate (although none of them approaches 24 hours of cooking).
It has always been an oversimplification to say that cooking time and cooking temperature are inversely proportional to each other—that the same, or similar, results can be obtained in a short time at a high temperature as for a longer time at a lower temperature. That concept is woefully inadequate, except over a very limited range of times and temperatures, because cooking is not a matter of simply injecting a given number of calories of heat into a food. As the old jazz song would have it, “It ain’t whatcha do, it’s the way hotcha do it.”
At the time of our discussion, the world was in one of its periodic energy crises, and Paula worried that long, low-temperature roasting might use more energy than shorter, higher-temperature roasting. Fascinated, I leapt at the challenge. Rather than taking the experimental approach, spending days in the kitchen after turning off all electrical devices in the house (it’s amazing how many there are, if you count them) except my electric oven and recording the readings on the electric meter, I decided to take the theoretical approach and try to solve the problem mathematically. Here’s what I came up with.
There are two energy-consuming stages in roasting meat: preheating the oven to the roasting temperature and maintaining that temperature during the roasting period.
It will obviously require more energy to preheat the oven to the higher of the two temperatures. (The actual difference in energy usage will depend on the characteristics of the individual oven.) But in either case the preheating time is short compared with the total roasting time, so we can probably neglect that difference. The difference in preheating times does, however, work in favor of less energy consumption by the low-temperature method.
During the roasting period, the oven will be persistently trying to cool down by losing heat to its surroundings. But whenever its temperature falls to a certain level, the oven’s automatic temperature control feeds in gas or electrical energy to replenish the heat that was lost. Thus, over the entire roasting period, the total energy input should be equal to the total energy lost by cooling. I could then obtain the energy usage under the two roasting conditions by calculating the rates of energy loss by cooling. The average rate of cooling (in calories per hour or Btu’s per hour) times the number of roasting hours should give me the total amount of energy used.
For my calculations I used Isaac Newton’s Law of Cooling (yes, that Isaac Newton), which says that the rate of cooling of a hot body is proportional to the difference in temperature between the body and its surroundings. In this case, the “body” is the air inside the oven, and its surroundings are the air in the kitchen. (The intervening oven walls slow the transfer of heat but don’t change the amount of heat that is ultimately transferred.)
Because all the heat-transfer parameters will differ from one case to the next, I can’t calculate absolute amounts of energy loss. But from Newton’s Law, I can calculate the break-even time: the number of slow-roasting hours at which the energy usage becomes equal to the energy usage in the fast-roasting method. If we slow-roast any longer than this, we will be using more energy than in the fast method.
Here are the results of my calculations. (Gluttons for mathematical detail may consult “(Warning: calculus ahead)” on chapter 9.)
In Paula’s first example, the energy break-even point for slow roasting at 180°F comes out to be about 9 hours. Thus, roasting for 24 hours at 180°F will use substantially more energy than roasting for 3 hours at 375°F. But 24 hours at 180°F is a rather extreme set of slow-roasting conditions anyway.
In Paula’s second example, the energy break-even point for slow roasting at 250°F comes out to be about 5 hours, which is close enough to Paula’s desired 6 hours. So go for it, Paula! The energy police will not break down your door.
What I’ve found, then, is that long, slow roasting need not use more energy than faster, higher-temperature roasting, provided that the slow roasting is not done at too low a temperature. Somewhere between 225 and 250°F (106 and 121°C) is probably the lowest practical limit. But if energy consumption isn’t an issue, by all means pull out the stops and cook your roast at any temperature above about 165°F (74°C) which is hot enough to kill most surface germs. Or do as Paula recommends in The Slow Mediterranean Kitchen: Blast or sear the surface of the meat first to take care of any surface germs before you lower the oven to roasting temperature.
Sidebar Science: (Warning: calculus ahead)
TO COMPARE the fast (f) and slow (s) methods of roasting a particular piece of meat to a given state of doneness, we will compare the total amount of oven cooling during fast roasting for hf hours at Tf degrees with the total amount of oven cooling during slow roasting for hs hours at Ts degrees.
To obtain the number of slow-roasting hours at which the two energy consumptions are equal, we’ll equate the two cooling rates and calculate hs , the energy break-even time for slow roasting.
For this application, Newton’s Law of Cooling can be written
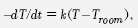
where T is the oven temperature, t is time, and Troom is the room temperature. The constant k depends on the specific oven and is assumed to be the same under both roasting conditions.
If the temperature fluctuations in the oven are relatively small compared with the oven temperatures themselves, and if the successive cooling periods are relatively short compared with the numbers of hours of roasting, we can approximate the differential rate of cooling with a temperature difference divided by a cooling time. Moreover, I will assume that the total amounts of time spent in cooling-and-reheating cycles under both sets of conditions are at least comparable. This can be partially justified by considering that the slow, low-temperature roasting, even though lasting longer, will require fewer reheating cycles because of its slower cooling rate.
Using these assumptions, we obtain
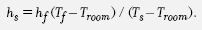
In words, the number of slow-roasting hours that consumes the same amount of energy as fast roasting is equal to the number of fast-roasting hours times the number of degrees above room temperature in the fast method, divided by the number of degrees above room temperature in the slow method. It doesn’t matter whether the temperatures are in Fahrenheit or Celsius, because only differences in temperature are involved.
In Paula’s examples, if the fast method roasts for 3 hours (hf = 3) at a temperature Tf =375, the energy break-even point hs for slow roasting at temperature Ts = 180 comes out to be 8.6 hours, and the energy break-even point for slow roasting at temperature Ts = 250 comes out to be 5.1 hours.
I can’t understand why Paula decided not to put these calculations in her book.
Paula Wolfert’s Slow-Roasted Leg of Lamb with Pomegranate Glaze and Red Onion–Parsley Relish
Low-temperature cooking delivers meltingly tender, rare meat. The lamb is first browned in a hot oven, then the temperature is reduced to 225°F. Roasting continues until the internal temperature of the meat reaches 130 to 135°F. The roast must rest before carving. The temperature will slowly rise to 135 to 140°F for a rare and juicy roast.
When carving, start at the shank end and slice perpendicular to the main bone. To obtain tender meat, always slice across the grain. Serve this Turkish-style lamb with the traditional Red Onion–Parsley Relish.
1 bone-in leg of lamb, 5 to 6 pounds
2 tablespoons pomegranate concentrate or molasses
1/3 cup water
1½ tablespoons extra-virgin olive oil
½ cup finely chopped onion
4 large cloves garlic, crushed
2 teaspoons tomato paste
1 teaspoon crushed red pepper flakes, preferably
Aleppo or Turkish
Pinch of sugar
Salt and freshly ground black pepper
1 cup chicken or vegetable broth
1 to 2 tablespoons unsalted butter
Red Onion–Parsley Relish (recipe follows)
1. Five to 6 hours before you plan to serve the lamb, trim off the excess fat, leaving about a ¼ -inch layer. In a large, deep bowl, dilute the pomegranate concentrate or molasses with the water. Stir in the olive oil, onion, garlic, tomato paste, red pepper, and sugar. Add the lamb and turn to coat. Let stand for no longer than 2 to 3 hours at room temperature, turning once or twice.
2. About 3 hours before serving, place a rack in the lower third of the oven. Preheat the oven to 450°F.
3. Set the lamb, fattiest side up, on a rack in an oiled shallow roasting pan. Season the lamb with plenty of salt and black pepper and set in the oven. Immediately reduce the oven temperature to 250°F. Roast the lamb, basting occasionally with the pan drippings, for 1¾ hours. Turn the roast over and continue roasting and basting for about 30 minutes longer, or until the lamb reaches an internal temperature of 130 to 135°F.
4. Remove the lamb to a carving board, cover loosely with foil, and let rest for 15 to 20 minutes. (During this time, the temperature will rise to 135 to 140°F.) Meanwhile, defat the pan juices. Add the broth, set the pan over medium heat, and stir to scrape up all the brown bits that cling to the bottom. Boil until reduced to napping consistency. Adjust the seasoning and keep hot.
5. Carve the lamb and serve with the sauce and the accompanying onion-parsley relish.
MAKES 6 TO 8 SERVINGS
Red Onion–Parsley Relish
2 red onions, thinly sliced
1 teaspoon coarse salt
½ cup chopped fresh flat-leaf parsley
1 teaspoon ground sumac*
Toss the red onions with the coarse salt. Rub the salt into the slices and let stand for 5 minutes. Rinse the onions under cold running water and drain thoroughly. Mix the onions with the parsley and sumac. Serve within 30 minutes.
MAKES ABOUT 1 CUP