CHAPTER 13
Dental Erosion
Ann‐Katrin Johansson, Inga B. Arnadottir, Göran Koch, and Sven Poulsen
Over the past decades the dental community has shown an increasing concern about erosive tooth wear, especially in children and adolescents. This anxiety is based on clinical observations, together with reports from many countries suggesting not only a high prevalence, but also a possible increase of both occurrence and severity of dental erosion. In the young individual the literature points to the global rise in soft‐drink consumption as the most significant factor in the development of dental erosion. Other factors such as lifestyle changes, a strong perception of the importance of body image for success, and chronic diseases are other possible reasons for an increasing prevalence of dental erosion. Tooth wear has a multifactoral etiology and is a result of concurrent action of different mechanisms and factors on the teeth in the oral environment. Dental erosion is one of these components and defined as a loss of tooth substance by a chemical process that does not involve bacteria. Besides erosion, dental attrition and abrasion may occur at the same or at different occasions, which adds to the complexity of the phenomenon of wear.
Studies have shown that without presoftening by acid of the dental hard tissue the wearing effect from attrition and abrasion is reduced greatly. Dental erosion is therefore regarded as the most important factor in the development of tooth wear and attrition, and abrasion considered to be of less significance [1]. Another term used in connection with dental wear is abfraction, a combination of erosion and stress in the buccal area of the cemento–enamel region provoking micro fractures (Box 13.1).
Prevalence
Recent studies claim a high and increasing prevalence of erosions in children and adolescents. Comparison of these studies is often difficult since the diagnostic criteria, scoring systems, and choice of teeth and surfaces to be scored vary. However, there are sufficient studies to give a general idea of the prevalence and distribution of erosion both in the primary and in the young permanent dentition (Table 13.1).
Table 13.1 Prevalence of dental erosion in children (primary teeth) and adolescents (permanent teeth) in different countries. Prevalence denotes erosive damage reaching the dentin or close to dentinal exposure of large surfaces
| Country | Age (years) | No. of individuals | Prevalence (%) | Authors (year) |
| Children | ||||
| UK | 4–5 | 178 | 30 | Millward et al. (1994) |
| UK | 5 | >1000 | 24 | Downer (1995) |
| UK | 1.5–4.5 | 1658 | 8 | Moynihan & Holt (1996) |
| Saudi Arabia | 5–6 | 354 | 34 | Al‐Majed et al. (2002) |
| Ireland | 5 | 202 | 21 | Harding et al. (2003) |
| India | 5–6 | 100 | 30 | Deshpande et al. (2005) |
| Germany | 2–7 | 463 | 13 | Wiegand et al. (2006) |
| Sweden | 5–6 | 135 | 13 | Hasselkvist et al. (2010) |
| Adolescents | ||||
| UK | 14 | 1035 | 30 | Milosevic et al. (1994) |
| UK | 15 | >1000 | 2 | Downer (1995) |
| Saudi Arabia | 20 | 95 | 16 | Johansson et al. (1996) |
| Cuba | 12 | 1010 | 17 | Kunzel et al. (2000) |
| Saudi Arabia | 12–14 | 862 | 26 | Al‐Majed et al. (2002) |
| Iceland | 15 | 278 | 22 | Arnadottir et al. (2003) |
| UK | 14 | 1308 | 13 | Dugmore et al. (2003) |
| UK | 14 | 2351 | 53 | Bardsley et al. (2004) |
| Turkey | 11 | 153 | 28 | Caglar et al. (2005) |
| Denmark | 15–17 | 558 | 1.6 | Larsen et al. (2005) |
| Sudan | 12–14 | 157 | 22 | El Karim et al. (2007) |
| Iceland | 12 | 757 | 16 | Arnadottir et al. (2010) |
| Iceland | 15 | 750 | 31 | Arnadottir et al. (2010) |
| Norway | 16–18 | 795 | 15 | Søvik et al. (2014) |
| Sweden | 13–14 | 227 | 12 | Hasselkvist et al. (2010) |
| Sweden | 18–19 | 247 | 22 | Hasselkvist et al. (2010) |
| Sweden | 20 | 494 | 18 | Isaksson et al. (2014) |
The prevalence of erosion involving dentin in children aged 2–7 years has been reported to vary from 8 to 34%, while erosion limited to the enamel is definitely more frequent. Most reported studies in the young permanent dentition are carried out in adolescents aged 11–20 years and the prevalence of erosion in dentin varies from 2 to 53%. Longitudinal studies indicate an increased number of teeth being affected by erosion and also an increase in severity with increasing age.
Distribution of dental erosion
The distribution of dental erosion is not uniform within the dental arches. Studies have shown that it is not possible to predict exactly the localization of such lesions depending on their etiology [2]. It is, however, clear that erosion will affect some teeth more than others. In children and adolescents, the maxillary anterior teeth, especially the palatal surfaces, and permanent first molars are those most often affected. The distribution of dental erosion in 12‐ and 15‐year‐old boys and girls are shown in Figure 13.1(a,b). The highest frequencies of erosion, both in boys and girls, were found in the mandibular first molars. The figures indicate an increased prevalence of erosion at age 15 compared with 12 years of age.
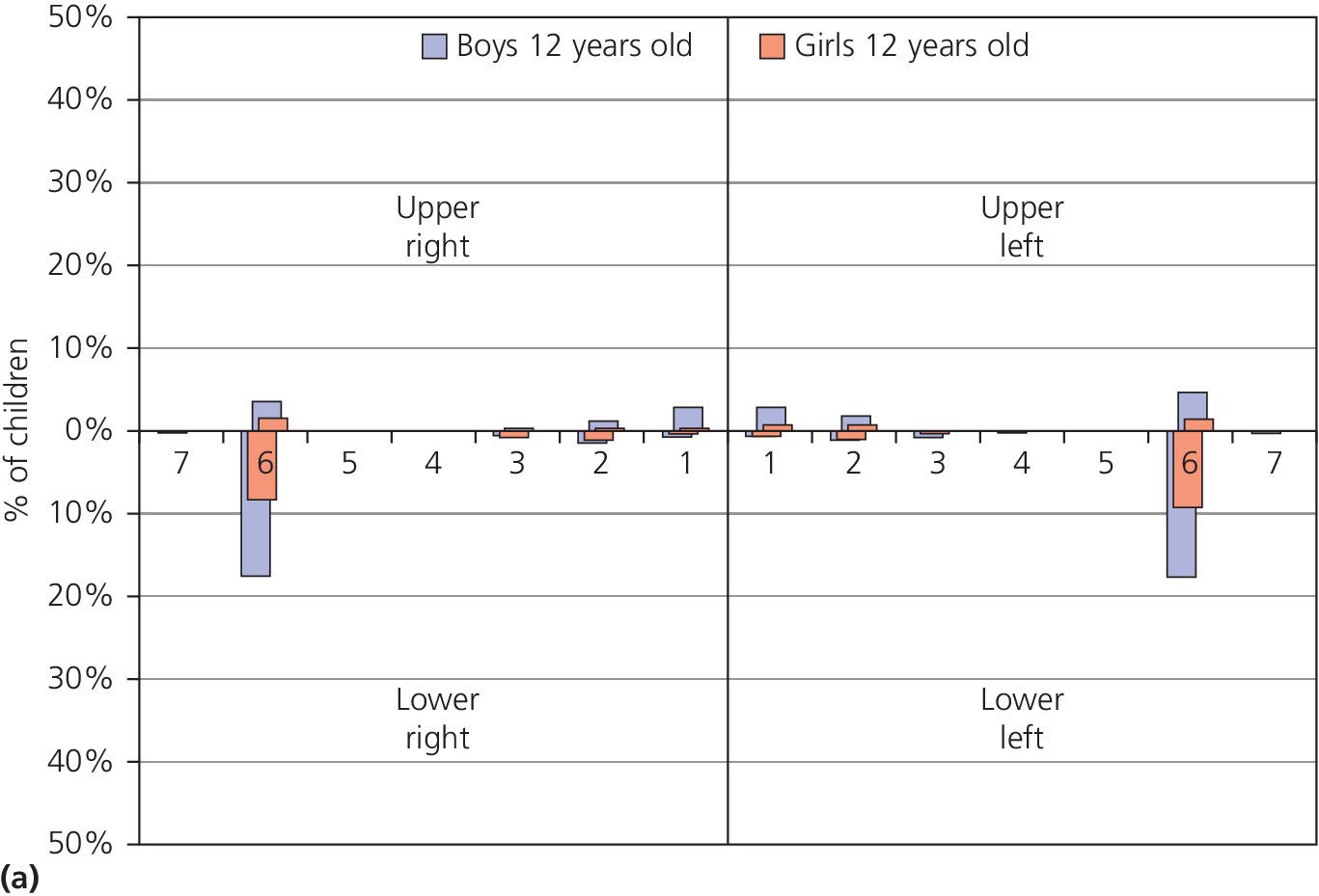
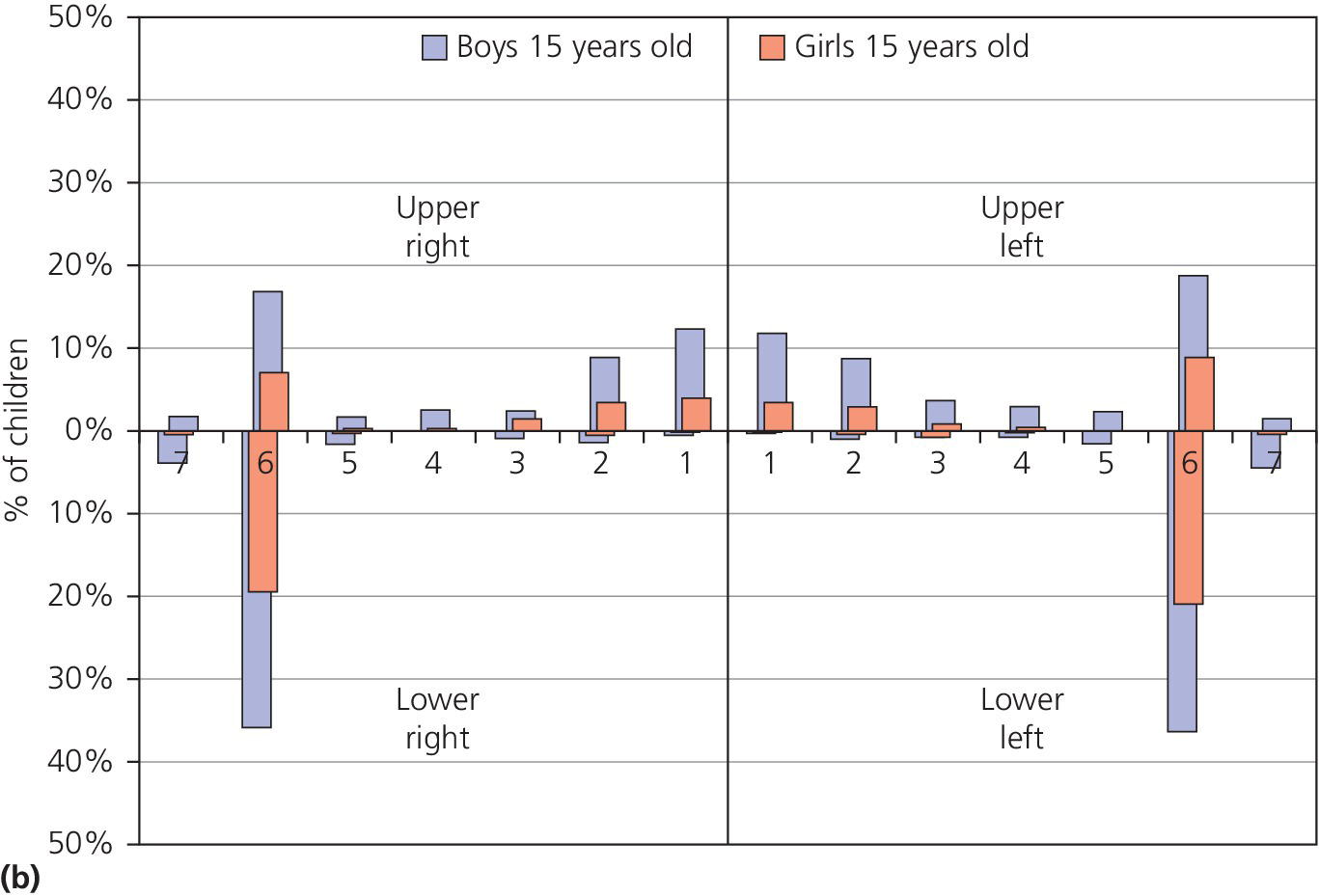
Figure 13.1 Distribution of dental erosion by tooth and by gender. Percentages of boys and girls with dental erosion by teeth at (a) 12 years of age and (b) at 15 years of age.
Source: Arnadottir et al. 2010 [46]. Reproduced with permission of John Wiley & Sons.
In severe cases of erosion in palatal surfaces of maxillary incisors a shoulder may be present cervically (Figure 13.2). Occasionally the approximal surfaces may also be affected. Cuppings (for explanation see second paragraph under heading “Clinical examination”) in the permanent dentition in children and adolescents are most commonly seen on the mandibular first molars, but can be found on all cusp tips or incisal edges (Figure 13.3). If erosion has been diagnosed on one tooth surface, it is important to examine other surfaces carefully for signs of tooth surface loss. This could, for example, be the presence of buccal–cervical defects or uprising restorations. Atypical localization of erosion exists and may be the result of, for instance, lemon sucking (Figure 13.4). Microhardness measurements have shown that deciduous enamel is softer than permanent enamel and that the progression of erosion is relatively more rapid than in permanent enamel. In addition, the fact that deciduous teeth are smaller in size further enhances the risk for complications by dental erosion [3]. (See Figure 13.5.)
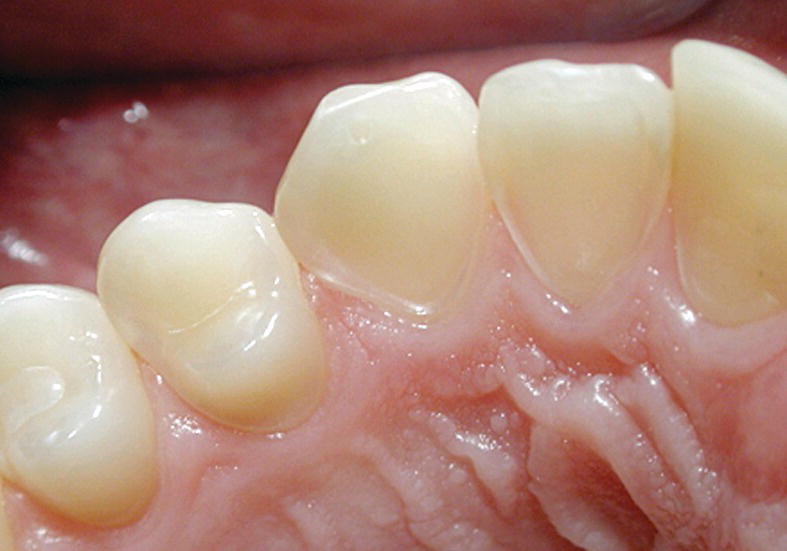
Figure 13.2 Severe erosion on the palatal surface of the upper incisors. The enamel close to the gingival margin is intact and a shoulder is shown.
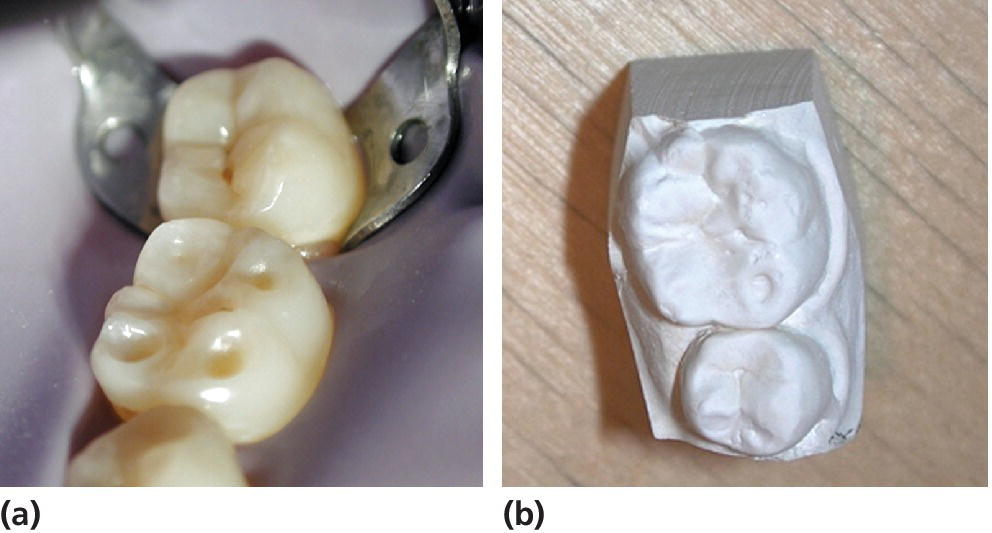
Figure 13.3 (a) Cuppings on a permanent first lower molar (tooth 36) in a young teenager. (b) Study cast from the same case.
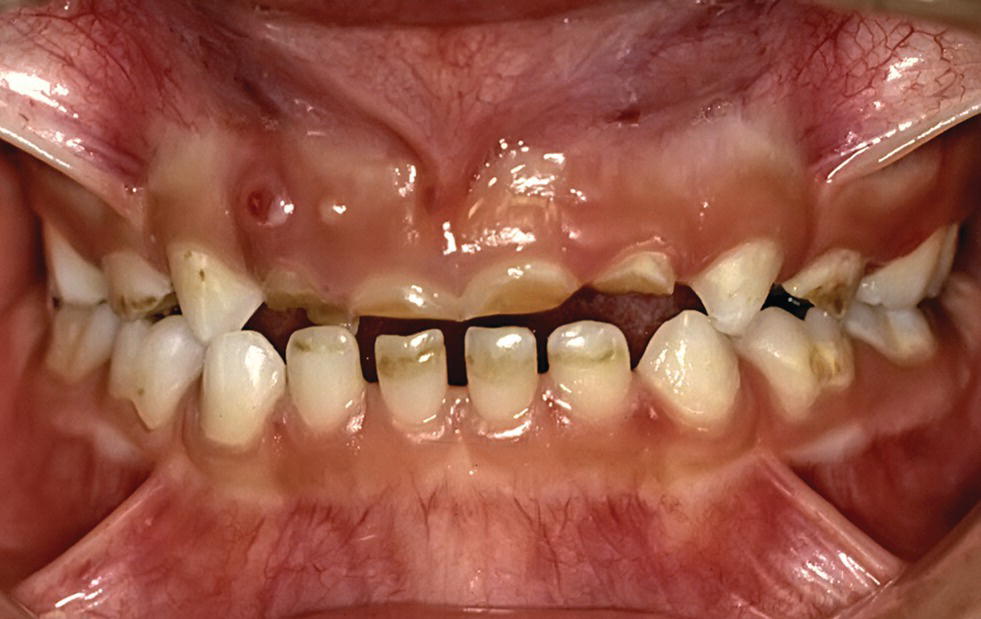
Figure 13.4 Dental erosion as a result of lemon sucking in a 6‐year‐old child. Dental erosion has caused severe endodontic problem especially in region 52 and 51.
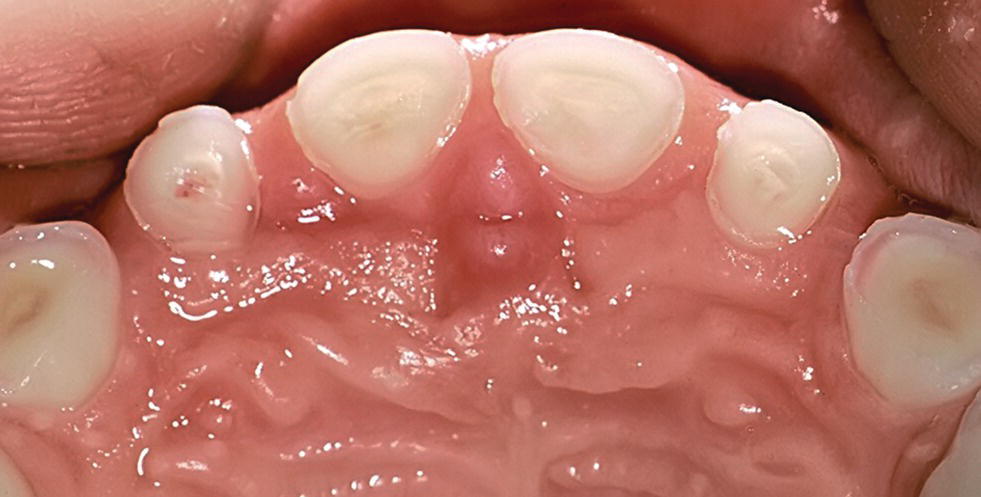
Figure 13.5 Near to pulp exposure on the palatal surfaces of anterior primary teeth. Secondary and tertiary dentin are shown.
Etiology
Traditionally, the etiologic factors for dental erosion have been divided into those of extrinsic and intrinsic origin (Box 13.2). The term “idiopathic erosion” has been used in cases of unknown etiology, but its clinical application is limited [4].
Examples of factors of extrinsic origins are all acidic drinks, and also acidic foods such as fruits and pickles, acidic medicines (e.g., acetylsalicylic acid and vitamin C tablets), and environmental exposure, such as highly chlorinated swimming pools.
The most common etiologic factor for dental erosion in children is high consumption of soft drinks, including fizzy drinks and fruit juices, which expose the dentition to frequent contacts with citric, phosphoric or malic acids. pH for some risk products are shown in Box 13.3.
Intrinsic factors include various gastrointestinal and eating disorders, e.g., vomiting, regurgitation, and rumination in which hydrochloric acid from the stomach comes into contact with the teeth.
Several chronic health conditions have been associated with dental erosion, especially those related to gastric acid/stomach disturbances and those affecting salivary secretion.
Factors decisive for erosive potential
The erosive potential of dietary components is estimated by measurements of initial pH, neutralizable acidity and ability to dissolve enamel in in vitro studies without the physiologic effects of saliva or frictional effects of the tongue. Besides the acidic challenges the content of calcium and phosphate in the products are important factors for the erosive potential and the dissolutions of tooth substance. Even at a very low pH it is therefore possible to prevent dental erosion in some drinks and milk products such as yogurt.
The interplay between these factors is often difficult to assess with regard to the overall risk and the clinical outcome.
Gastroesophageal reflux (disease)
Studies have shown that children with gastroesophageal reflux (disease) [GER(D)] have a higher prevalence of dental erosion compared to healthy children albeit it is considered to be a normal physiologic condition among infants. In the younger child, the symptoms will be diffuse and frequently described merely as stomach pain or persistent coughing. Symptoms in the older child have a more precise symptomatology: pain from the upper part of the stomach, heartburn, regurgitation, dysphagia, and coughing, especially at nighttime, are examples. In some cases, the patient does not present any typical symptoms at all; this is referred to as “silent reflux.” However, this condition may be detected by 24‐h pH monitoring, which is the gold standard in GER(D) diagnosis. In rare cases, reflux can be voluntary, which is called “rumination,” and most often found in connection with disabilities or eating disorders.
Asthma
Asthmatic problems are common in the child population and many asthmatic children have GER(D). While some reports have shown an increased prevalence of dental erosion in children with asthma others have not. In an asthmatic patient, bronchodilating medication may reduce salivary secretion and relax the lower esophageal sphincter with an increased potential for acidic reflux [5]. In addition, the medication itself may be acidic and mouth breathing resulting in dryness of the mouth is common, all of which raises the risk of dental erosion.
Eating disorders
Children and adolescents with eating disorders have an increased risk for erosion. In one study dental erosion was more than eight times more common in patients with eating disorders compared to controls. Self‐induced vomiting is seen in patients with bulimia nervosa, but also occurs in patients with other types of eating disorders; anorexia nervosa for example. Besides this, a high intake of sugar‐free soft drinks and fruits, excessive oral hygiene often combined with gastrointestinal disturbances, and salivary impairment are frequent among patients with eating disorders and raise the risk of erosion [6,7].
A large number of medications cause a dry mouth, e.g., anticholinergic drugs, medicines for GER(D), and psychiatric disorders. Cancer therapy with cytostatics or radiation treatment (often combined with vomiting) frequently compromises the function of the salivary glands and increases the risk for erosion. Diabetes (both types 1 and 2), hypoparathyroidism and hyperparathyroidism, hyperthyroidism, and congenital agenesis of salivary glands, Down syndrome, cerebral palsy, Prader‐Willi syndrome or the use of methylenedioxymethamphetamine (ecstasy) are other conditions associated with dental erosion [8].
Other factors associated with dental erosion
Drinking habits
An individual’s method of drinking acidic drinks plays an important role in the development of erosive lesions. The erosion damage is likely to be greater in those individuals who retain the drink in their mouth for some time before swallowing, compared to those who do not, as the contact time between the acid and the teeth is increased [9]. In a study, 43% of children with dental erosion were found to have a drinking habit such as swishing or holding compared to 3% in caries‐free and 15% in caries‐active controls [10], and in another study it was shown that the intraoral pH after drinking soft drinks differed between those with dental erosion and controls [11]. Dental erosion has also been associated with the consumption of acidic drinks in a baby’s bottle at bedtime or naptime [12]. Sucking through a straw may reduce the erosive potential of the drink if the straw is positioned towards the back of the mouth, which may be very difficult, especially for a child [13] (Figure 13.6).
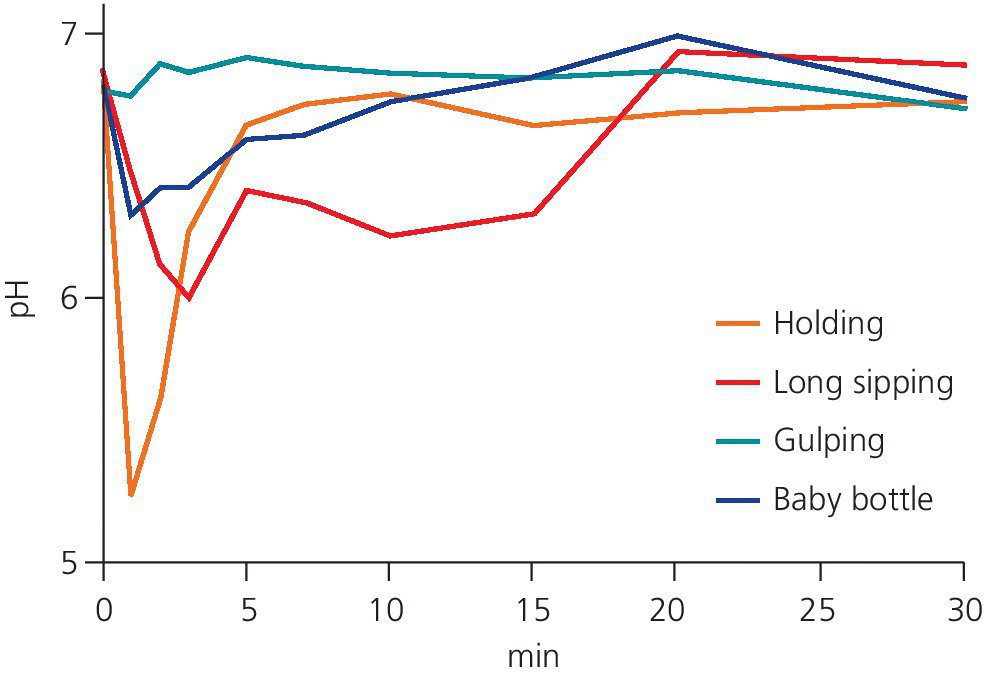
Figure 13.6 pH decrease (mean values) for three methods of drinking and nipping from a baby’s bottle using the microtouch method and Cola Light. Holding = holding the drink in the mouth for 2 min. Long sipping = sipping from a glass for 15 min. Gulping = swallowing quickly three times over 5‐min intervals. Baby bottle = nipping from a baby’s feeding bottle for 15 min.
Source: Johansson et al. 2004 [9]. Reproduced with permission of John Wiley & Sons.
Salivary factors
An increased erosive action may be expected if the salivary secretion rate and buffering capacity are reduced. Saliva is important in the prevention of erosions through dilution and clearance of acidic products, and plays an important role in pellicle formation and demineralization and remineralization. It has been stressed that salivary characteristics may represent a risk for developing dental erosion lesions. The thickness of the pellicle varies between individuals and between different locations in the mouth, and may be reduced by acidic challenges such as soft drinks. A thicker pellicle provides more protection than a thinner one, and it has been suggested that salivary secretion and pellicle thickness strongly influence the location and development of erosion [14–16]. When measuring unstimulated and stimulated salivary flow rates, buffering capacity, and number of mutans streptococci in three groups of children, one with extensive erosions, one without caries, and one with high caries prevalence, the following were found: the erosion group showed great similarities concerning caries with the caries‐free group and great similarities concerning saliva characteristics with the high caries group [17]. Similar results were found in another study where children with erosion showed lower unstimulated and stimulated salivary rates, lower buffering capacity, and lower salivary pH, and a larger maximum pH drop after consumption of soft drinks compared to children without both erosion and caries [18] (Box 13.2).
Oral hygiene practices
It has been shown that individuals with erosion have better oral hygiene than those without erosion [19]. The modern practice of cleaning teeth is certainly more erosion conducive than a more irregular, less methodical, and less vigorous method of oral hygiene, since the erosive lesion develops on surfaces free of plaque. A plaque‐free surface could be a result of oral hygiene activities, but also a consequence of natural cleaning from the lips, tongue, and cheeks. Approximal surfaces are seldom free of plaque and this may be the reason why they rarely develop erosive lesions.
Oral hygiene activity, after an acidic challenge on the tooth surface, has been discussed but might result in a larger loss of tooth substance due to its being “presoftened” by acids. It has therefore been suggested to delay toothbrushing for at least an hour after the acidic challenge.
Lifestyle
The view has been expressed that “the lifestyle of today seems to increase the acidic challenge to the dentition and thereby introduces a new risk factor for the dentition” [20]. Lifestyle has a large impact on both general and oral health. It varies over time and often reflects social circumstances including dietary habits, physical activity, drug usage, and stress‐related factors. Important changes in the modern lifestyle are the increase in soft‐drink consumption, the decrease in milk consumption, and reduced physical activity in association with an increasing prevalence of obesity, diabetes, osteoporosis, and being overweight in the child population [21–24].
The fact that most soft drinks are consumed by young children and adolescents [25] especially increases the risk of dental erosion in these groups. Adolescents who frequently consume sport drinks during and after exercise when salivary secretion is lowered [15], as well as the young computer user who stays awake at night with the help of a cola drink, will have an increased risk of dental erosion.
A healthy lifestyle may also have an impact on the occurrence and severity of dental erosion, e.g., vegetarianism and dieting practices which are common among vegetarian children and adolescents. Vegetarians and children on a diet often have a high consumption of juices, fruits, and vegetables, perhaps with an added vinegar dressing, and in order to be orally healthy they also perform very thorough tooth hygiene.
Another recent lifestyle trend, especially among girls, is an increasing, unhealthy fixation on body shape and appearance. This may cause psycho‐pathological behavior leading for example to anorexia and/or bulimia including starving, vomiting, and a food pattern promoting dental erosion. These severe cases should be treated by a physician and at the same time by a dentist to ensure that the dental erosions do not aggravate [7].
The occurrence of dental erosion does not follow any obvious socioeconomic pattern. While some reports show that erosive wear is more common in children and adolescents with low socioeconomic status, other reports demonstrate the reverse. The same applies to gender differences, but in general it seems that boys are more affected than girls, which could be explained by the fact that boys generally drink more soft drinks compared to girls of a similar age.
The consequences of a new lifestyle often become obvious only when it is firmly established and it is difficult to revert to earlier routines. Thus, the oral medical aspects of dental erosion involve not only a proper dietary and medical history, but also knowledge of the person’s lifestyle. For example, it has been shown that the number of times per week children drink soft drinks relates to the number of teeth with dental erosion (Figure 13.7) [26].
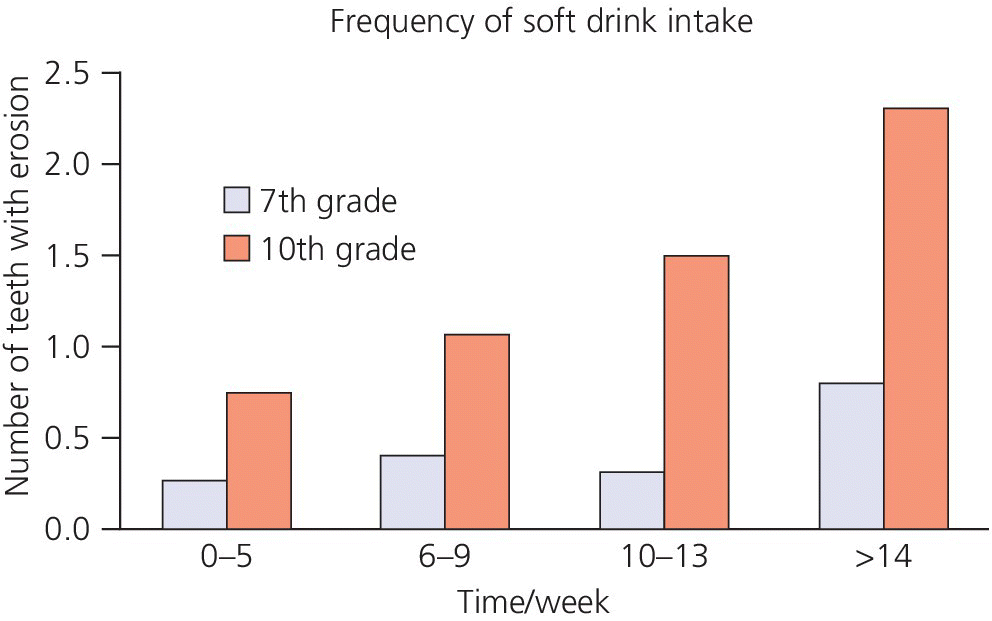
Figure 13.7 Dose‐response relationship between frequency of soft drink intake and number of teeth affected by dental erosion in 7th grade (12 year‐old) and 10th grade (15 year‐old) children.
Source: Arnadottir et al. 2008 [26]. Reproduced with permission of Icelandic Dental Journal.
Case history and clinical examination
Case history
Children and adolescents are exposed to many different erosive challenges which may result in initiation or aggravation of already present dental erosion lesions. Taking a case history in patients with dental erosion is therefore often both extensive and time consuming. In younger children, where the case history has to be obtained from the parents, the dentist should recognize that not all parents are fully aware of their children’s dietary and drinking habits. Information about general health, e.g., gastric conditions, eating disorders, and asthma, including the type of medication, is relevant. Besides the obvious need for a dietary history, with special reference to acidic intakes including consumption of acidic drinks and fruits, other lifestyle factors which may be associated with an increase of exposure to erosive agents also need to be addressed. These include sports activities, computer use, and oral hygiene habits, for example. A dietary history should be performed over several days, preferably a week in order to include the weekend. This can, of course, be difficult especially in younger children, who besides their own family may meet different persons during the day in daycare centers and schools as well as when being with friends or grandparents. Consideration needs not only to be given to when and to what exposure, but also in what way, such as the method of drinking. Information on oral hygiene habits should be obtained. Finally, it is important to remember that an erosive lesion may not be the result of the child’s present habits, but could be caused by earlier exposure to an erosive challenge such as a previous period of high consumption of soft drinks. Some practical steps in taking a case history and in the clinical assessment of dental erosion are listed in Box 13.4.
Clinical examination
The first step in the clinical examination of a patient for dental erosions is a visual inspection of the tooth surface. This should be completed in good operating light and on a clean and dry tooth surface. Since the erosive process may cause dental sensitivity varying from slight sensitivity to severe pain, any air blow has to be performed carefully. A larger mirror, such as the type used for occlusal views in intraoral photography, is preferable during the examination of erosion since the enlarged view allows an inspection of several teeth at the same time [27].
The clinical manifestations of erosion are easy to miss in the early stage as there is no discoloration of the lesion, no sticking on probing, and often no or only limited symptoms. In the advanced stages the clinical signs of erosion are more obvious. If the morphology of the tooth surface has changed, not due to mechanical forces or dental caries, dental erosion should be suspected. In cases of severe tooth wear, erosion should always be regarded as a possible underlying factor since the softening of the dental hard tissue by the erosive process may increase the loss of tooth surface. The surface of erosive lesions can have a different appearance. It can be shiny or matte, may be irregular, but is often rounded or flat, as if “melted.” The tooth’s developmental structures may vanish and in more severe cases the macromorphology is altered. In children, the many macroscopic structures observed in the newly erupted tooth will be lost. This results in a typical glasslike feeling of a smooth surface upon careful and gentle probing with a sharp probe, and is an important sign of dental erosion. Small concavities can sometimes be seen on the buccal surface of the tooth. In cases with extensive dentine exposure, “jagged” enamel edges can be seen. The pulp may, in severe cases, be visible through the remaining tooth substance: this is most common on the palatal surface of the maxillary central deciduous teeth. There may also be a formation of clinically detectable protective tertiary dentin, or direct pulpal involvement. One common clinical sign of erosion in both permanent and primary dentition is cuppings: a “peephole” in the enamel, most frequently seen on the cusp tips on the first molars [28] (Figure 13.8). Two or more cuppings may fuse together to form one larger affected area on the occlusal surface [21]. Restorations may sometimes be seen rising above the tooth surface as a result of dental erosion. These are not only visible clinically, but may also be detected on radiographs, study casts, and photographs.
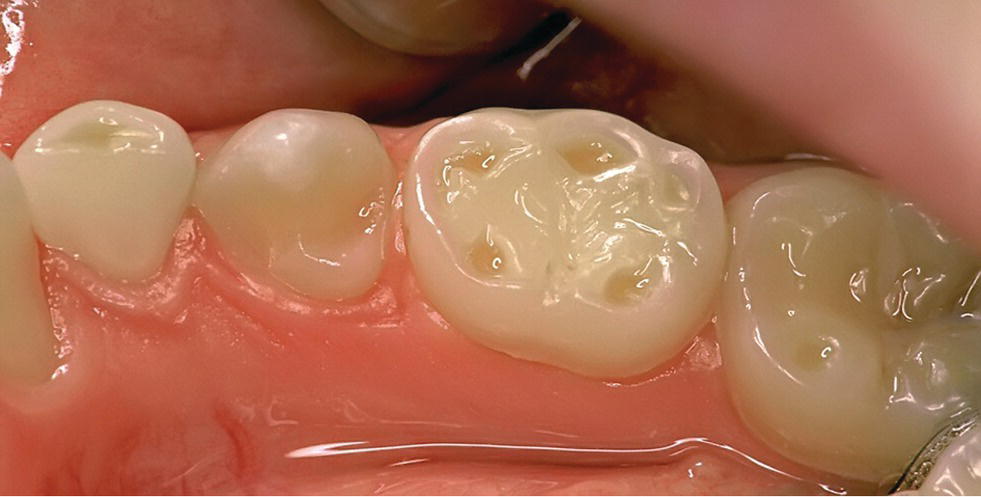
Figure 13.8 Clinical signs of erosion: cuppings on primary molars (teeth 83 and 85) and permanent molar (tooth 46).
Besides the more obvious loss of tooth substance resulting in esthetic and/or functional problems during eating or drinking, the process may also result in malocclusion. The mechanisms behind this are compensatory alveolar growth as a result of loss of tooth substance. Some clinical characteristics are listed in Box 13.5. (See Figure 13.8.)
Study casts are valuable in visualizing the three‐dimensional aspects of changes affecting morphological features, but should carefully be prepared to enhance the diagnostic possibilities. The angle at which a cast is viewed will affect the appearance of the lesions. Intraoral photographs are of some value in the clinical assessment, but the two‐dimensional image, poorer lightning conditions, and improper angling of the camera strongly limit its value as a diagnostic tool.
Salivary secretion rate, of both stimulated and resting saliva, as well as analyses of buffering capacity, should be considered.
Indices for dental erosion
Different index systems for grading dental wear and dental erosion in the clinical situation have been described. Some systems are based on partial recordings of marker teeth [28,29], while others recommend full mouth scoring or combinations thereof [30,31]. A combined index for clinical and epidemiologic purposes with a few marker teeth may be practical: four and six registrations per child has been proposed in permanent and deciduous teeth, respectively, including grading of cuppings to simplify data collection [28,32].
A few indices are designed only for deciduous teeth [30] or only for permanent teeth [33], while others have been used for both deciduous and permanent teeth [28,29]. Generally, in most situations, a relatively simple ordinal scale is suitable. A combined approach of grading erosive wear using clinical inspection, examination of study casts and photographs, offers advantages compared to systems which rely on a single or a dual approach. When choosing a scoring system it is important to consider the type of investigation for which the data will be used, e.g., clinical grading for management purposes, population‐ or laboratory‐based research. In the clinical setting, the duration of the examination needs to be kept in mind since some of the systems are very time consuming to use with respect to both patients and clinicians. In order to perform a more thorough registration of the severity of dental erosion as well as follow‐up, a scale that distinguishes between different stages of erosion is necessary.
Early enamel erosion is found commonly in both children and adolescents. These lesions have to be detected early in order to optimize the possibility for prevention of further damage. A grading system for dental erosion must be able to distinguish between different stages of enamel erosion as well as severe erosion into dentin or close to dentinal exposure. The system with marker teeth, based on the most common localization of dental erosion, is timesaving both on an individual basis and in epidemiology (Figure 13.9a,b). If the scale for grading erosion is too coarse, only the more severe erosive lesions can be recorded, which increases the risk for overlooking early signs of dental erosion, losing the possibility of early intervention and follow‐up. Thus, the choice of marker teeth and the scale used for registration is essential in both clinical and epidemiologic work. Box 13.6 shows a scale used for grading dental erosion in children and adolescents on buccal and palatal surfaces on upper anterior teeth. Additional registrations should include recording of cuppings on first permanent or deciduous molars and of buccal cervical defects. A separate scale for registration of cuppings in primary molars and permanent first molars is shown in Box 13.7.

Figure 13.9 Illustrations of different severities of dental erosion graded according to the scale in Box 13.6: (f) is a study model of the patient in (e) illustrating that intraoral photographs could be combined with grading of study models as the two complement each other in assessment of dental erosion.
Source: Johansson & Carlsson 2006 [47]. Reproduced with permission of Gothia Fortbildning.
Follow‐up of patients with erosions
Frequently, the etiologic, modifying and/or aggravating factors of dental erosion will persist throughout life. Therefore, a child once diagnosed with dental erosion should be monitored and supervised for a long period. Besides providing the patient and parents with the necessary information about the condition and prophylactic advice, it is possible to use photographs, serial study casts, salivary analysis, and consultations with the patient’s family physician (e.g., in cases of gastric problems, eating disorders, or asthmatic problems) as tools to control dental erosion.
The progression of erosion may be assessed by comparing study casts taken over an appropriate time interval. In some cases radiographs, preferably bitewings, may be of value. Also, it is generally important to predict the longevity for the involved tooth and the impact of this for the patient. One exception to this rule is severely affected deciduous teeth just before exfoliation. Studies have shown that the risk of severe erosion in the permanent dentition increases nearly four times if severe erosion is present in the deciduous dentition [34]. The clinical consequence of a diagnosis of dental erosion should be considered on an individual basis since the same severity of erosion may be perfectly acceptable in one patient but not in another. If a child or adolescent is diagnosed with erosion it is more likely that this process is active than if an erosive lesion of the same severity is diagnosed in an adult. As a permanent tooth in a younger individual has a longer life expectancy than in an adult, the same erosive problem may therefore have a higher risk of complications over time in younger compared to older people.
In order to give the patient a proper diagnosis, the clinical documentation of dental erosion is essential. The modern electronic dental file system available on the market today has built‐in functions suitable for registration of dental caries but should be seldom for tooth wear/erosion, which complicates its clinical recording.
Prevention
General aspects
Control of dental erosion can be achieved both by individual counseling of patients, and by general, population‐targeted interventions. In both strategies, the competency of the dental team in recognizing the early stages of erosion as well as understanding its pathogenesis has been strongly emphasized [27]. Of equal importance is effective communication as regards dental erosion to the patient, the parents and the general population, since the success of preventive measures in most cases will depend strongly on their proper understanding of the condition and their cooperation.
Counseling of patients
If a patient is suspected of suffering from progressive dental erosion it is important to hasten the investigation in order to repress or prevent further tooth surface loss. As mentioned earlier, the resistance to erosive challenges varies considerably between individuals. Therefore, no generally valid recommendations can be given concerning how much acidic drink a specific child can drink without risk of erosion. However, for a child with diagnosed dental erosion it is clear that the acidic challenges during some period of its life have been too severe. It may well be that the signs of erosion can be linked to earlier habits or conditions and less to present circumstances. Prevention of dental erosion is often complicated, and before any recommendation is decided on it is advisable to evaluate the progression of erosion.
In cases of erosion due to extrinsic causes, reduction of all acidic intakes is one method of preventing further erosion. This may require lifestyle changes, and may, therefore, at times involve not only the affected child but the whole family. If a proper diagnosis of erosion in the deciduous dentition can be made and the factors involved understood, a preventive strategy for the permanent dentition is possible since the same etiologic factors will most likely also affect the child’s permanent dentition [3]. Advice and information about erosion at the right time may in some patients totally prevent further progression, while in others the situation may be more difficult to control. Examples of recommendations in cases of extrinsic dental erosion are given in Box 13.8.
The ultimate goal of prevention is to avoid the initiation of any erosive lesions and to stop progression of existing lesions. In some patients with active erosion the preventive measures may only result in a slowing of the progression. This is especially the case in patients with dental erosion due to intrinsic causes such as GER(D) and eating disorders, conditions which may be found in both children and adolescents and most often difficult to control.
One way to eliminate or reduce intrinsic factors for erosion is to consult the patient’s physician. By treating or controlling the general disease, the most effective prevention of further dental erosion is obtained. In patient with GER(D), a proper medical examination, sometimes including 24‐h pH measurement of the acidity in the esophagus, and then diagnosis and treatment with medication or surgery is of importance. In cases of nocturnal reflux, the head of the child’s bed could be raised 5–10 cm in order to reduce the risk of the stomach’s acidic content reaching the oral cavity. If antacid is used as a medication for gastric reflux this could be kept in the mouth before swallowing in order to neutralize any acid in the mouth [35]. Dietary advice should, as always, be given with due consideration to the medical recommendations. If an eating disorder is suspected, contact with parents, school nurse, physician, or psychologist/psychiatrist is suggested. Dietary advice in patients with eating disorders should be given in collaboration with medical or psychiatric personnel as it has to consider not only dental and medical health but also psychiatric health.
As long as the affected tooth substance is not completely lost and if sufficient calcium, phosphate, or fluoride is available on the surface, remineralization of newly eroded surfaces may be achieved. Rinsing the mouth with water, on the other hand, when there are acidic challenges will stop the erosive process but does not lead to remineralization. If the brittle organic network that remains on the tooth surface immediately after an erosive challenge is worn off by abrasion or attrition, the possibility of remineralization is lost. Therefore, it is recommended that toothbrushing with toothpaste should be avoided for at least an hour after an acidic challenge. Toothbrushing without toothpaste, after an erosive challenge, has shown deposition of salivary components, inducing remineralization [36]. Dental erosion has also been found to be associated with brushing before sleeping and after meals, and with the type of toothbrush and the brushing technique [18,37].
Salivary secretion rate is very important in order to prevent dental erosion and needs to be increased when insufficient. First, ensure that the intake of liquid is adequate, and second, salivary secretion can be stimulated using fluoride tablets or special sugar‐free tablets. Chewing gum can also be recommended but it should be used with caution as chewing gum may increase occlusal wear in some patients. However, some studies have shown that remineralization of early erosion may occur after chewing calcium‐containing chewing gum. If medication causes reduced salivary secretion or increased reflux, or is acidic itself, elimination or substitution of the drug should be considered in collaboration with medical expertise. Examples of recommended oral hygiene habits for patients with erosion are given in Box 13.9.
In patients with nocturnal bruxism the use of an occlusal protective splint can be suggested in order to eliminate abrasion or attrition. However, there has been speculation that the splint may act as a reservoir for acid during nocturnal regurgitation, thus increasing the risk of dental erosion.
The clinical evidence for the use of fluoride in the control of dental erosion is weak [38]. However, fluoride therapy is recommended in patients with erosion even though the effect of the treatment is limited. At the same time it has to be stressed that a change of lifestyle, resulting in a reduction of etiologic factors, is much more effective than any other preventive measures.
Sensitivity due to erosion can be reduced in different ways. However, it has to be strongly emphasized that the first and most important action is to eliminate or reduce all acidic challenges as far as possible. In combination with this, intensive fluoride therapy may be useful, as well as the use of special toothpastes. Other methods could be to block the enlarged dentinal tubules with different types of bonding or desensitizer. This treatment may reduce the sensitivity, but the prognosis depends on the longevity of the blocking agent and its resistance towards further erosive challenges in the mouth. It might, therefore, be necessary to repeat the treatment. In severe cases it can even be necessary to restore the eroded surfaces in order to prevent or eliminate sensitivity and further erosive damage. In some cases fissure sealants may be helpful, e.g., in the prevention and restoration of cuppings. Box 13.10 shows examples of relevant clinical measures in cases of erosion.
Population‐based prevention
On a population basis it is likely that the most effective prevention of erosion would be to reduce the intake of acidic drinks among children and adolescents both with and without dental erosion. The preventive effect of information about dental erosion is also valuable even though knowledge does not always lead to a change of lifestyle. Studies have shown that the availability of soft drinks in school vending machines is an important factor in the choice of beverage. Campaigns promoting a healthier lifestyle among children and adolescents have mainly targeted being overweight or obese, but have also been of value for the improvement of oral health and the prevention of dental erosion.
Other appropriate preventive measures are the modification of acidic drinks by the addition of calcium. Such modified products are available in some countries in order to reduce the erosive potential of the drink [39].
Restorative treatment
There are a number of indications for restorative interventions in cases of dental erosion. Restorations can be performed to improve esthetics and function, or to manage sensitivity and pain, which does not respond to other preventive actions. In some instances, restorative intervention is needed in order to prevent undesirable orthodontic effects or further loss of tooth substance. However, all reconstructions have a limited longevity, which implies that a future treatment need exists for teeth that have been restored at an early age in children and adolescents. It is equally important to recognize that tooth surface loss does not always need to be restored and that any decision on restorative intervention always should be individually based.
Soft drinks, especially fruit juices, have been proven to reduce the longevity of many dental materials. After a year of immersion in vitro in apple and orange juice, conventional glass ionomers have dissolved completely, but were less affected by cola drinks. Microhardness was reduced on resin‐based glass ionomers and compomers during immersion in cola, but was affected less than by fruit juice [40]. Studies have shown that even though many dental materials will be affected by acid, resulting in different types of problems, composite materials and ceramics seem to have a good stability [41,42]. The effect of soft‐drink consumption on the bonding between orthodontic appliance and tooth surface has been investigated in one combined in vivo and in vitro study. It was shown that children who were exposed to soft drinks several times daily had poorer retention of their brackets than children who were not [43]. Another study indicated that the longevity of posterior restorations, with direct and indirect composites, worn by erosion is shorter than control teeth restored due to dental caries. The failure rate after three years was 50% in the erosion group and 20% in the control group. Twenty‐eight percent of the restorations in the erosion group were completely lost and another 22% were fractured during this period of time [44].
In children and adolescents with dental erosion, composites have to be considered as the first choice for restorative treatment even though the long‐term performance in cases of dental erosion has not been sufficiently evaluated. In children with erosive wear it is preferable that restorative treatment is performed using a minimally invasive technique. If the retention of the restoration needs to be reinforced, as in the case of restorations of cuppings, it is recommended that a bevel preparation is used. There is an increased risk for progressive wear in a cupping since any acidic challenges may remain inside the cupping thus resulting in a prolonged contact time between the acid and the tooth surface.
In patients with localized worn anterior teeth, the loss of vertical height caused by the erosion may cause a problem with space for the restorative material. Raising the bite using the Dahl technique or a modified Dahl technique can in many cases be a good solution [45]. A modified Dahl technique may be performed by using an occlusal splint which is used night and day for a number of months. But the most common modification of the Dahl technique used today, especially on young individuals, is a build‐up on the maxillary anterior teeth including palatal surfaces. In any case the Dahl technique will create a posterior open bite bilaterally, but with time occlusion will be normalized by a compensatory alveolar growth of posterior teeth as well as intrusion of anteriors and in case of composite restorations the bite will be normalized (Figure 13.10).
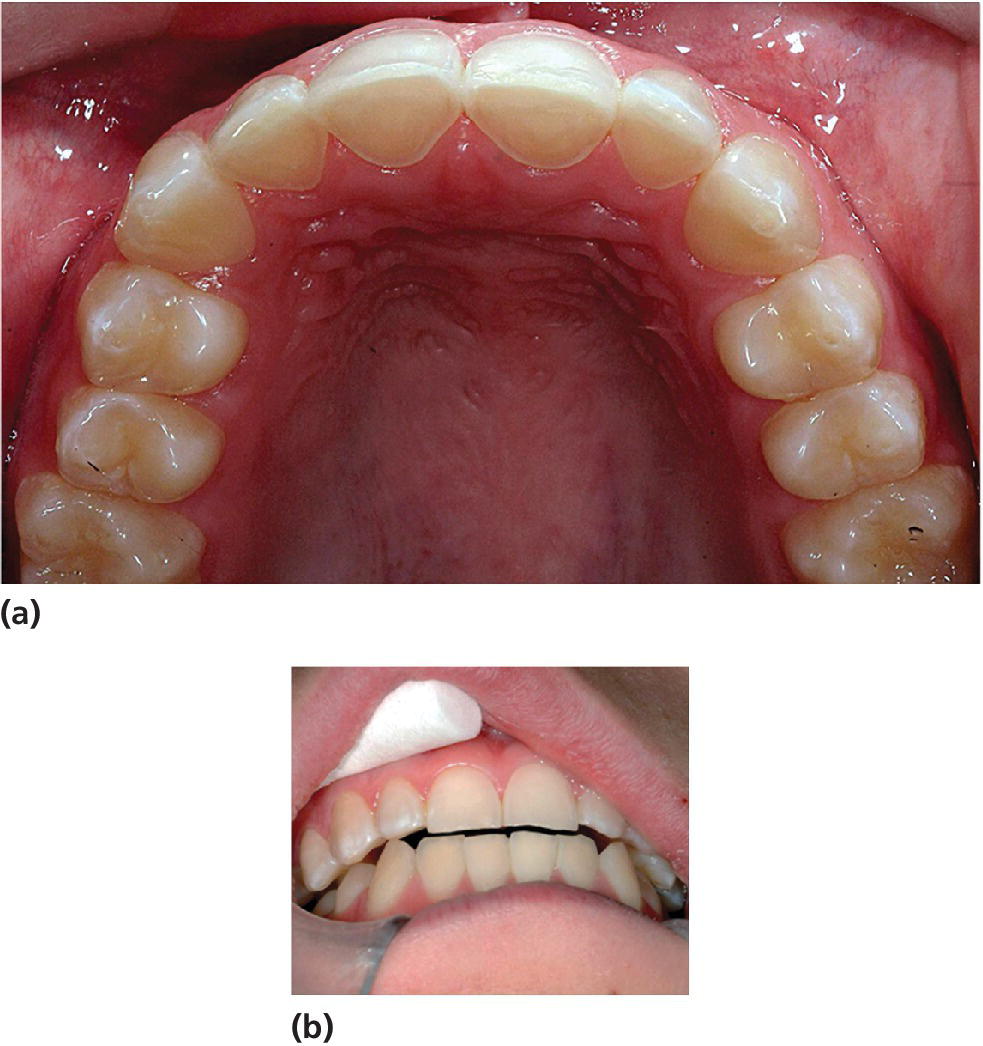
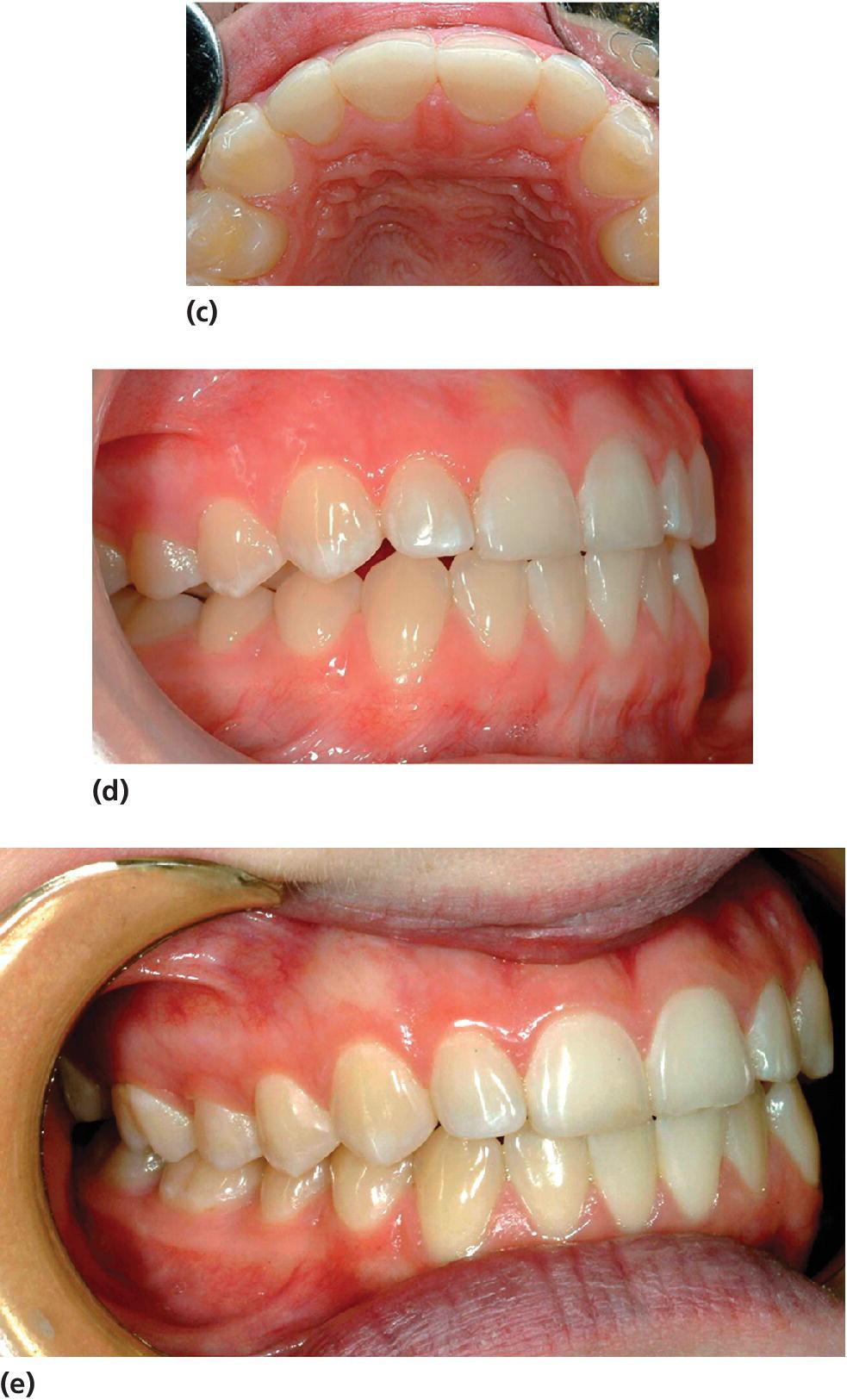
Figure 13.10 (a) Fifteen‐year‐old girl with palatal erosion and sensitivity on maxillary anterior teeth. (b) Composite restoration took place on tooth 12, establishing a new vertical dimension sufficient for restoring 11, 21, and 22 (c). (d) As a result, posterior disocclusion is present which, however, is normalized after approximately 4 weeks due to (e) compensatory eruption and alveolar growth.
Source: Johansson & Carlsson 2006 [47]. Reproduced with permission of Gothia Fortbildning.
Occasionally, prosthetic therapy is the choice of treatment even in young individuals or adolescents.
It has to be remembered that restorative treatment is not causal therapy of the disease. The erosion process can still be active, affecting the dentition, and the patient will be in need of future support and follow‐up.
References
- 1. Bartlett DW. The role of erosion in tooth wear: aetiology, prevention and management. Int Dent J 2005;55 (Suppl 1):277–84.
- 2. Järvinen V, Rytomaa I, Meurman JH. Location of dental erosion in a referred population. Caries Res 1992;26:391–6.
- 3. Johansson AK, Sorvari R, Birkhed D, Meurman JH. Dental erosion in deciduous teeth—an in vivo and in vitro study. J Dent 2001;29:333–40.
- 4. Imfeld T. Dental erosion. Definition, classification and links. Eur J Oral Sci 1996;104:151–5.
- 5. Shaw L, Al‐Dlaigan YH. Childhood asthma and dental erosion. ASDC J Dent Child 2000;67:102–6.
- 6. Öhrn R, Enzell K, Angmar‐Månsson B. Oral status of 81 subjects with eating disorders. Eur J Oral Sci 1999;107:157–63.
- 7. Johansson AK, Norring C, Unell L, Johansson A. Eating disorders and oral health: a matched case‐control study. Eur J Oral Sci 2012;120:61–8.
- 8. Young WG. The oral medicine of tooth wear. Aust Dent J 2001;46:236–50.
- 9. Johansson AK, Lingstrom P, Imfeld T, Birkhed D. Influence of drinking method on tooth‐surface pH in relation to dental erosion. J Oral Sci 2004;112:484–9.
- 10. O’Sullivan EA, Curzon ME. A comparison of acidic dietary factors in children with and without dental erosion. ASDC J Dent Child 2000;67:186–92.
- 11. Moazzez R, Smith BG, Bartlett DW. Oral pH and drinking habit during ingestion of a carbonated drink in a group of adolescents with dental erosion. J Dent 2000;28:395–7.
- 12. Al‐Malik MI, Holt RD, Bedi R. The relationship between erosion, caries and rampant caries and dietary habits in preschool children in Saudi Arabia. Int J Paediatr Dent 2001;11:430–9.
- 13. Edwards M, Ashwood RA, Littlewood SJ, Brocklebank LM, Fung DE. A videofluoroscopic comparison of straw and cup drinking: the potential influence on dental erosion. Br Dent J 1998;185:244–9.
- 14. Young WG, Khan F. Sites of dental erosion are saliva‐dependent. J Oral Rehabil 2002;29: 35–43
- 15. Amaechi BT, Higham SM, Edgar WM, Milosevic A. Thickness of acquired salivary pellicle as a determinant of the sites of dental erosion. J Dent Res 1999;78:1821–8.
- 16. Sorvari R, Rytömaa I. Drinks and dental health. Proc Finn Dent Soc 1991;87:621–31.
- 17. O’Sullivan EA, Curzon ME. Salivary factors affecting dental erosion in children. Caries Res 2000;34:82–7.
- 18. Sánchez GA(1), Fernandez De Preliasco MV. Salivary pH changes during soft drinks consumption in children. Int J Paediatr Dent 2003;13:251–7.
- 19. Johansson AK, Lingström P, Birkhed D. Comparison of factors potentially related to the occurrence of dental erosion in high‐ and low‐erosion groups. Eur J Oral Sci 2002;110:204–11.
- 20. Imfeld T, ten Cate JM. Preface. Eur J Oral Sci 1996;104:149.
- 21 American Academy of Pediatrics Committee on School Health. Soft drinks in schools. Pediatrics 2004;113:152–4.
- 22. Hasselkvist A, Johansson A, Johansson AK: Association between soft drink consumption, oral health and some lifestyle factors in Swedish adolescents. Acta Odontol Scand 2014;72:1039–46.
- 23. Harnack L, Stang J, Story M. Soft drink consumption among US children and adolescents: nutritional consequences. J Am Diet Assoc 1999;99:436–41.
- 24. Isaksson H, Birkhed D, Wendt LK, Alm A, Nilsson M, Koch G. Prevalence of dental erosion and association with lifestyle factors in Swedish 20‐year olds. Acta Odontol Scand 2014;72:448–57.
- 25. Rugg‐Gunn AJ, Nunn JH. Diet and dental erosion. In: Nutrition, diet and oral health. Oxford: Oxford University Press, 1999.
- 26. Arnadottir IB, Gudlaugsson JO, Jonsson SH. Body Mass Index (BMI) caries and erosion in Icelandic teenagers. Icelandic Dental Journal 2008;26:46–9.
- 27. Johansson AK. On dental erosion and associated factors. Swed Dent J Suppl 2002;(156):1–77.
- 28. Johansson AK, Johansson A, Birkhed D, Omar R, Baghdadi S, Carlsson GE. Dental erosion, soft‐drink intake, and oral health in young Saudi men, and the development of a system for assessing erosive anterior tooth wear. Acta Odontol Scand 1996;54:369–78.
- 29. Downer MC. The 1993 national survey of children’s dental health. Br Dent J 1995;178:407–12.
- 30. O’Sullivan EA. A new index for the measurement of erosion in children. Eur J Paediatr Dent 2000;1:69–74.
- 31. Bartlett D, Ganss C, Lussi A. Basic Erosive Wear Examination (BEWE): a new scoring system for scientific and clinical needs. Clin Oral Investig. 2008;12 Suppl 1:S65–8.
- 32. Hasselkvist A, Johansson A, Johansson AK. Dental erosion and soft drink consumption in Swedish children and adolescents and the development of a simplified erosion partial recording system. Swed Dent J 2010;34:187–95.
- 33. Lussi A, Schaffner M, Hotz P, Suter P. Dental erosion in a population of Swiss adults. Community Dent Oral Epidemiol 1991;19:286–90.
- 34. Ganss C, Klimek J, Giese K. Dental erosion in children and adolescents—a cross‐sectional and longitudinal investigation using study models. Community Dent Oral Epidemiol 2001;29:264–71.
- 35. Meurman JH, Kuittinen T, Kangas M, Tuisku T. Buffering effect of antacids in the mouth—a new treatment of dental erosion? Scand J Dent Res 1988;96:412–17.
- 36. Kuroiwa M, Kodaka T, Kuroiwa M, Abe M. Brushing‐induced effects with and without a non‐fluoride abrasive dentifrice on remineralization of enamel surfaces etched with phosphoric acid. Caries Res 1994;28:309–14.
- 37. Al‐Dlaigan YH, Shaw L, Smith AJ. Dental erosion in a group of British 14‐year‐old, school children. Part III: Influence of oral hygiene practises. Br Dent J 2002;192:526–30.
- 38. Austin RS, Stenhagen KR, Hove LH, Tveit AB, Moazzez RV, Bartlett DW. The effect of single‐application fluoride treatment on simulated gastric erosion and erosion‐abrasion of enamel in vitro. Int J Prosthodont. 2014;27:425–6.
- 39. Hooper S, Hughes J, Parker D, Finke M, Newcombe RG, Addy M, et al. A clinical study in situ to assess the effect of a food approved polymer on the erosion potential of drinks. J Dent 2007;35:541–6.
- 40. Aliping‐McKenzie M, Linden RW, Nicholson JW. Effects of pH on the microhardness of resin‐based restorative materials. Oper Dent 2005;30:661–6.
- 41. Jaeggi T, Gruninger A, Lussi A. Restorative therapy of erosion. Monogr Oral Sci 2006;20:200–14.
- 42. Mohamed‐Tahir MA, Tan HY, Woo AA, Yap AU. The effect of Coca‐Cola and fruit juices on the surface hardness of glass‐ionomers and ‘compomers’. J Oral Rehabil 2004;31:1046–52.
- 43. Oncag G, Tuncer AV, Tosun YS. Acidic soft drinks effects on the shear bond strength of orthodontic brackets and a scanning electron microscopy evaluation of the enamel. Angle Orthod 2005;75:247–53.
- 44. Bartlett D, Sundaram G. An up to 3‐year randomized clinical study comparing indirect and direct resin composites used to restore worn posterior teeth. Int J Prosthodont 2006;19:613–17.
- 45. Dahl BL, Krogstad O, Karlsen K. An alternative treatment in cases with advanced localized attrition. J Oral Rehabil 1975;2:209–14.
- 46. Arnadottir IB, Holbrook WP, Eggertsson H, Gudmundsdottir H, Jonsson SH, Gudlaugsson JO, Saemundsson SR, Eliasson ST, Agustsdottir H. Prevalence of dental erosion in children: a national survey. Community Dent Oral Epidemiol 2010;38:521–6.
- 47. Johansson AK, Carlsson GE. Dental erosion: bakgrund och kliniska aspekter. Förlagshuset Gothia, 2006.