
I f there is a flaw in our genetic makeup, the most straightforward way to address the problem is to correct the flaw. This project is not new. Measures aiming at improving the hereditary endowment of humanity were proposed in the nineteenth century under the name of “eugenics,” literally the pursuit of good genes.
This term was conceived, with the project it implies, by a cousin of Darwin, Sir Francis Galton (1822–1911), who initiated intelligence testing and defended the view that psychological characteristics are hereditary and that society should defend itself against undesirable traits by selective breeding, if not by stronger measures against the genetically unfit. First favorably received in certain conservative and racist quarters of Victorian England and the United States, eugenism prompted a number of discriminatory measures, condemned today, with as summit of ignominy the horrors of the Nazi regime. Since then, anything smacking of genetic discrimination has become socially and politically abhorrent.
The most shocking aspect of the original eugenic project is the distinction it made between bearers of “bad genes” and bearers of “good genes,” and, even more, the means it proposed to eliminate the former or, at least, prevent them from procreating and so propagating their flaws. But another form of eugenics that does not suffer this objection can be envisaged today; it aims at eliminating or correcting certain unfavorable genes we all have in common, genes that were once adaptive, even essential to our survival, but have become increasingly maladaptive. If these genes are now dysfunctional although retained by natural selection, perhaps we should use our brains to correct them before they do us in. We may well have the means to do so, at least potentially.
It started in 1996 in Scotland, at the Roslin Institute, near Edinburgh, where a small team headed by Ian Wilmut first succeeded in cloning a sheep, the now world-famous Dolly. Since then, the procedure has been successfully repeated on more than a dozen different mammalian species, including mice, rats, cows, pigs, horses, cats, and dogs. Success with humans has been claimed but not substantiated. What is the procedure?
First, what does “cloning” mean? Derived from the Greek klon, twig, the noun “clone” is defined as a group of organisms formed asexually from a single ancestor. Bacteria typically develop as clones. So can some plants, starting from a single cutting. Until Dolly, however, mammals had never been cloned. Actually, calling Dolly a clone is not strictly correct, as Dolly is an individual, not a group. But the word is now sanctioned by usage and refers to a nuclear-genomic “carbon” copy of an individual obtained without sexual reproduction.

Fig. 15.1. Cloning by nuclear transfer, as compared to fertilization. On the right, the normal mechanism of fertilization: (1) two gametes, oocyte and spermatozoon, each with a single genome (haploid), join to form (2) a totipotential fertilized egg cell possessing two genomes (diploid) and a cytoplasm supplied almost exclusively by the oocyte (see fig. 5.5). On the left, nuclear transfer: (3) an unfertilized oocyte is enucleated; (4) a somatic (body) cell with two genomes (diploid) is enucleated; (5) the diploid nucleus of the somatic cell is inserted into the enucleated oocyte, to produce a cell with a diploid, differentiated nucleus and the oocyte’s cytoplasm; (6) the differentiated nucleus is “deprogrammed” under the influence of the oocyte’s cytoplasm and is returned to a totipotential state. Either cell is now ready for implantation.
On paper, the method whereby this result is achieved is simple, but it is very delicate in practice (fig. 15.1). One takes an unfertilized oocyte, removes its nucleus, and replaces it with the nucleus of a differentiated cell, from skin, liver, or mammary gland, for example. The renucleated oocyte is then implanted into a female body, where, under proper stimulation and if all goes well, it develops into a normal individual with a nuclear genome identical to that of the donor of the inserted nucleus, as in an identical twin, except that, unlike the identical twin, the individual would have a different mitochondrial genome (which comes from the oocyte). This quasi-identical twin of the owner of the transferred nucleus is what we now call a clone.
Note the change in chromosome number caused by this procedure. We saw in chapter 5 that oocytes, like spermatozoa, have a single set of chromosomes; they are haploid. When an oocyte and a spermatozoon join in fertilization, they give rise to a diploid cell, with two sets of chromosomes, one set from the oocyte, one set from the spermatozoon. All the other cells of the body derived from the fertilized egg are likewise diploid, with the exception of germ cells, which undergo reduction to a haploid state in the course of their maturation. Cloning, therefore, means replacing the haploid nucleus of an unfertilized oocyte with the diploid nucleus of some body cell; it is a means of providing an oocyte with a diploid nucleus without fertilization (see fig. 15.1). For the rest, it involves the same manipulations as are widely applied in the procedure known as in vitro fertilization (IVF).
A key difference between nuclear transfer and fertilization lies in the potential of the nucleus. In fertilization, the resulting diploid nucleus is totipotential; it can, by specific activation of certain genes and silencing of others, give rise to any of the differentiated nuclei characteristic of the different cell types found in the body. This is what happens in the course of the successive cell divisions whereby the fertilized egg de velops into a complete organism. In cloning, this totipotential nucleus is replaced by the already fully committed nucleus of some differentiated body cell. One might expect to see such a renucleated egg cell multiplying into a clone of the cell that provided the nucleus. Surprisingly, this is not what happens. Under the influence of the oocyte cytoplasm, the developmental clock turns back to zero in the transplanted nucleus, which undergoes “deprogramming” to the totipotential state. As a result, the inserted nucleus recovers its lost youth.
This astonishing return to virginity by a transplanted nucleus was first observed in the 1970s, in amphibians, by the British biologist John Gurdon, who is the true “father” of animal cloning. Shortly afterward, a Swiss investigator claimed to have accomplished the same result on mice but later had to retreat in some confusion—perhaps unjustly in view of what is now known of the hazards of the technique—because his findings could not be reproduced. As a consequence of this unfortunate incident, mammalian cloning fell into disrepute, until Dolly was produced by Wilmut and his team, who obviously belonged to those who “didn’t know it couldn’t be done and so went ahead and did it.”
Cloning can be used for several purposes (fig. 15.2). One form, called reproductive cloning, aims at creating a younger, quasi-identical twin of the donor of the transplanted nucleus. This method can be used, for example, to perpetuate valuable stock.
In so-called therapeutic cloning, the resulting early embryo is sacrificed, to provide totipotential stem cells that are kept for possible future use in the repair of damaged tissues in the donor of the nucleus. The advantage of this technique is that, because of their genetic kinship with the cells of the donor, such stem cells are not immunologically rejected by the host, as would be the case with foreign grafts.
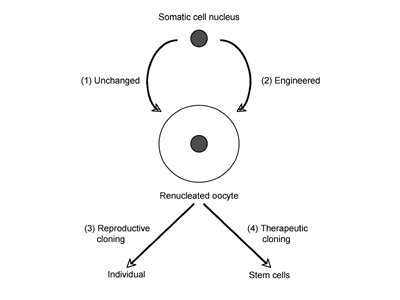
Fig. 15.2. Different kinds of cloning. The somatic cell nucleus destined to be transplanted into the enucleated oocyte is inserted as such (1), or after modification by genetic engineering (2). The renucleated oocyte is either implanted to develop into an individual (3, reproductive cloning) or used for the generation of stem cells (4, therapeutic cloning). Combination of steps 2 and 3 is used to create genetically modified (transgenic) animals.
In a particularly important form of cloning, which could be called engineering, the transplanted nuclei have been subjected to some genetic modification, leading to the creation of genetically modified organisms (GMOs), also called “transgenic.” This procedure is now widely used in research, where it has allowed a host of important discoveries. There are also many industrial applications of this technique, used, for in stance, to generate animals that manufacture valuable human proteins in their milk or to endow animals with new, useful properties, such as the ability to subsist on new kinds of foods, enhanced productivity, or lower harmfulness to the environment. Thus, pigs have been equipped with a bacterial enzyme that renders the animals able to digest an important phosphorus-containing component of their food that is normally excreted unbroken and is a major source of pollution of streams and lakes in the neighborhood of pig farms, where it favors eutrophication, an excessive proliferation of algae that stifles other forms of life. These animals have been called “enviropigs” because of their beneficial effect on the environment. Genetically modified plants are also produced on a large scale, by a different procedure, which I describe in chapter 18.
So far, no authenticated instance of human reproductive cloning has been reported. But this is due mainly to ethical constraints that most countries impose on such attempts. There is no reason to suspect that the technology would not be applicable to human cells. Certainly, this possibility has already inspired innumerable conjectures and debates. Creating a younger copy of oneself or replacing a lost child have been cited as possible applications of human reproductive cloning, a procedure that is, at present, prohibited by most legislatures around the world.
Human therapeutic cloning is allowed in many countries but fiercely opposed in others, including the United States, where prolife advocates condemn the destruction of a potential human embryo inherent in the technique. Because of this opposition, an immense research effort is aimed at obtaining stem cells without cloning. Possible sources are umbilical cord blood or, if some genetic commitment is acceptable, differentiated tissues, most of which are now known to contain pluripotential (though not totipotential) stem cells. Special hope is put in a recently achieved procedure claimed to turn back the clock in a fully differentiated cell by the modification of only four genes.
As to engineering cloning, its envisaged human applications are restricted to the medical correction of some specific gene defect already identified. The use of this technique to produce “designer babies” has, however, been much evoked, hotly discussed, and mostly rejected.
Production of designer babies would require considerable improvement of present cloning methods. In today’s state of the art, the percentage of failures remains too high to allow an ethically acceptable use of human material. The number of “failed” embryos and their fate would create unmanageable situations. Should safe procedures someday become available and should the ethical ban against human reproductive cloning be lifted, this technique could conceivably be used to engineer genetically modified humans, by knocking out or otherwise mutating certain genes or by inserting new ones.
The main problem, if such manipulations were to become possible and ethically acceptable, would be the choice of the genes to be manipulated or inserted. We would need to know much more than we know now about the genetic control of human qualities. This is a major unsettled issue. Experts even disagree on the extent to which certain characteristics are genetically determined, let alone agreeing on more specific relationships. It does, however, seem probable that complex psychological traits or talents do not depend on single genes. It is unlikely that one could engineer the production of a new young Mozart or Einstein or Martina Navratilova by simple genetic manipulation.
Even if the necessary knowledge of genetics should one day be available, one still would have to agree on what qualities to aim for. If some central authority were in charge, the threat of a Brave New World would loom. In order to avoid this dangerous difficulty, some have proposed formation of a genetic “supermarket,” in which prospective parents would choose the qualities of their offspring à la carte. This idea may seem grotesque, but it could arguably be viewed as an improvement over the present lottery, in which the genetic makeup of children is decided by pure chance among a huge number of combinations of parental genes. Remember that, because of the genetic reshuffling that takes place in the course of germ cell maturation, oocytes and, especially, spermatozoa come in a large number of different genetic varieties. The genome of a fertilized egg depends on whichever of the millions of spermatozoa contained in an ejaculate succeeds in entering the available oocyte first. Substituting reasoned choice for such a blind game could be seen as desirable.
Whether such manipulations will ever be attempted or accomplished cannot be predicted at the present time. They certainly are not among the measures that can be contemplated today as a solution to the present pressing problems of humankind. Even if one knew what to do and how to do it, the question would remain: Who should benefit from the improvement? Changing six billion individuals hardly seems feasible. Starting with a small group destined to become the new Herrenvolk is too reminiscent of Nazism to even be thinkable today. Letting selection be based on the ability to pay for the very expensive procedures involved, as proposed by the American biologist Lee Silver in his book Remaking Eden (1997), would be consistent with laissez-faire, though not with most people’s concept of democracy or social fairness. It will be up to future generations to make the appropriate decisions if they are ever able to produce viable genetically modified humans. The problem does not presently exist, but it may well some day.