30 Vertebral Augmentation in Instrumented Spinal Fusions
John W. Amburgy, Patrick R. Pritchard, Steven M. Theiss, James Mooney, D. Mitchell Self, and M. R. Chambers
Summary
Vertebral augmentation is a minimally invasive treatment known to reduce pain and disability and improve the quality of life in patients with painful vertebral fractures. In addition to its applications across a wide range of etiologies, it offers promise in select patients when used in combination with instrumented fixation to help alleviate spinal instability. These patients often have elevated risks of hardware failure, collapse of constructs, and junctional fractures. Supplemental vertebral augmentation of pedicle screw fixation may mitigate these risks. Biomechanical studies demonstrate improved pullout strength of augmented screws placed into osteoporotic bone.
Keywords: vertebral augmentation, spinal fusion, pedicle screws, spinal fixation, burst fracture, osteopenia, osteoporosis
30.1 Introduction
The utility of vertebral augmentation is quite broad with applications ranging from the treatment of complications of benign disease such as osteoporosis to primary malignancies and metastatic disease. An important application is its use in combination with instrumented fixation in patients with spinal instability. These patients are a heterogeneous population but they have in common the need for bony vertebral fusion. They often have elevated risks of hardware failure, collapse of constructs, and junctional fractures. Supplemental vertebral augmentation of pedicle screw fixation offers the spine surgeon intraoperative options to mitigate these risks.
30.2 Instrumentation in Spinal Fusion
Roy-Camille is credited with the first description of posterior plates with screws positioned sagittally through the pedicles and articular processes with a system that he and Judet had been using since 1963.1 This became the foundation for pedicle screw fixation. Harrington and Tullos described the first transpedicular screw placement in the United States in 1969. Subsequent pedicle screw systems and modifications came from Louis, Steffee, Magerl, Luque, Wiltse, and others. Design modifications continue today in an effort to improve purchase, strength, and fusion outcomes.2–5
In 1991, Lorenz et al prospectively evaluated 68 patients with at least 6 months of disabling back pain who had failed conservative care to compare single-level posterolateral lumbar fusions (PLF) with and without pedicle screws. Twenty-nine patients were fused without hardware and 39 were fused with variable screw placement (VSP) fixation. An improved fusion rate was reported for patients undergoing pedicle screw fixation with pseudarthrosis seen in 58.6% of the non-instrumented group and no pseudarthrosis in instrumented patients. Pain improvement was better in the instrumented group (76.9 vs. 41.4%) and the rate of return to work was higher in the instrumented patients (72 vs. 31%) compared to the non-instrumented patients.6
In a report to the contrary, Thomsen et al suggested that pedicle screw fixation did not affect functional improvement or fusion rates long-term.7 Despite this controversy, pedicle screw fixation has become routine in posterolateral fusions. Guidelines for the performance of fusion procedures for degenerative disease of the lumbar spine were published in the Journal of Neurosurgery Spine in 2014. An association, but no direct correlation, was reported between increased fusion rates and pedicle screw and rod fixation as assessed with dynamic radiographs. Despite the routine nature of this practice, the guidelines recommend “the use of pedicle screw fixation as a supplement to PLF (posterolateral fusion) be reserved for those patients in whom there is an increased risk of nonunion when treated with only PLF.”8
Although an in-depth analysis of the addition of pedicle screw fixation to fusion constructs is not the focus of this chapter, one must understand that attempts to further stabilize the pedicle screw may be inextricably linked to data correlating pedicle screw fixation to improve the stability of the fusion and therefore the functional and radiographic outcomes. As seen in the examples above, results are not definitive.
30.3 Vertebral Augmentation in Spinal Fusion
Data supporting vertebral augmentation and its benefit to spinal fusion date back over 30 years. In 1986, Zindrick et al18 assessed various biomechanical performances of sacral screw fixation, including fixation augmented with polymethyl methacrylate (PMMA). The authors reported that placement of PMMA around a loosened screw restored fixation and doubled its pullout force. This was reproduced in osteoporotic models of the lumbar spine by Soshi et al9 who measured the pullout force of a 7-mm pedicle screw on normal cadaveric lumbar vertebrae compared to those with mild and severely osteoporotic bone. A pullout force of 1,056.4 N was required in the normal, non-osteoporotic group, 495.6 N in the mildly osteoporotic group, and 269.5 N in the severely osteoporotic group. The use of bone cement for augmentation reduced the risk of screw pullout in the osteoporotic vertebrae by a twofold increase in pullout strength.9
Mermelstein et al further categorized the bending moments of pedicle screws augmented with calcium phosphate (CP) cement. The bending moment was reduced by 59% in flexion and 38% in extension, and the mean stiffness was increased by 40%.10 Sarzier et al reported in 2002 that augmentation of osteoporotic vertebrae increased pedicle screw pullout forces. The maximum attainable force was approximately twice the pullout force of the non-augmented pedicle screws for each osteoporotic (Jikei scale) grade. The mean increase in pullout force in osteoporotic spines was 181% for Grade I, 206% for Grade II, and 213% for Grade III.11
30.4 Indications
Vertebral augmentation is used to treat vertebral compression fractures (VCFs) across a wide range of etiologies. Augmentation of spinal instrumentation may be indicated for a select patient population, including osteoporotic patients, those requiring revision surgery, especially those high-risk patients who may not tolerate lengthy general anesthesia and surgery, and adults with spinal deformity. In osteoporotic patients, augmentation may reduce the risk of screw pullout. In addition, patients who require revision of their instrumented spinal fusion should benefit from augmentation as it increases the stiffness and pullout strength of the replacement screws. Augmentation in high-risk patients can be used to provide construct stability and prolonged pain relief. Finally, augmentation has been shown to decrease the need for revisions and reduce the risk of proximal junctional fractures (PJFs) following instrumentation in adults with spinal deformities.12,13 See ▶Fig. 30.1.
30.5 Outcomes and Variables
Many variables affect outcomes following instrumented fusion, with or without augmentation. Here, we review outcomes of fusions with vertebral augmentation as they relate to etiology, method of delivery, instrumentation, cement composition, volume, and timing.
30.5.1 Osteoporosis
Significant risk of screw pullout and fractures exist in osteoporotic patients as coercive attempts at deformity correction are made with instrumentation. The risk of hardware failure is proportional to corrective forces applied. In osteoporotic patients, bone quality becomes most important in determining outcomes and the axiom, “bone holds metal; metal doesn’t hold bone” applies. Pedicle screw augmentation with PMMA improves the initial fixation strength and fatigue strength of instrumentation in osteoporotic vertebrae. Numerous studies of the lumbar and thoracic spine, ilium, and sacrum demonstrate a 1.5- to 2-fold increase in pullout strength of augmented screws compared to non-augmented screws.9,11,14–16 This benefit may be realized only in low-quality bone of osteopenic and osteoporotic patients.17
30.5.2 Revision Surgery
Similar to results in osteoporotic patients, PMMA or other cements can salvage screw fixation and increase the force required for pullout twofold.18 The pullout strength of augmented replacement screws in thoracolumbar vertebrae returned to baseline or increased above baseline in these revision cases.19,20 Initial and final stiffness of the larger diameter screws was also increased with the addition of the cement augmentation.21
30.5.3 Select High-Risk Patients
Comorbid disease or poor general health may preclude internal fixation and fusion surgery. In these select patients, vertebral augmentation may provide an alternative. For example, Puri and Erdem22 described two patients with multiple myeloma who had failed posterior spinal interbody fusions and had significant pain, but were felt to be at high risk of complications from general anesthesia due to their multiple comorbidities. Rather than lengthy construct revisions under general anesthesia, unilateral transpedicular vertebroplasties were performed under intravenous conscious sedation.
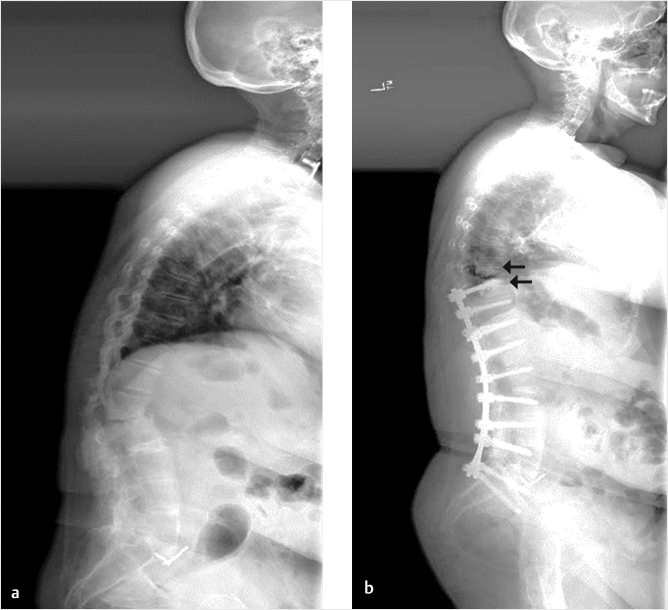
Fig. 30.1 (a, b) Sixty-five-year old female who underwent an instrumented fusion from T10 to the pelvis with a Smith Peterson osteotomy at L1–L2. Along with the instrumentation, she had polymethyl methacrylate (PMMA) placed in the upper instrumented vertebra (UIV) and the vertebra immediately cephalad to the construct (UIV+1) (black arrows in b). (This image is provided courtesy of Dr. Steven M. Theiss, MD.)
The first patient had previously undergone thoracic vertebrectomy with interbody device placement and dorsal internal fixation from T6 to T10. Six months later, when recurrent pain prompted MR and CT imaging, construct failure and apparent loosening of the interbody device was identified. Believing that constant micromotion by the cage might be causing the pain, the authors performed transpedicular vertebroplasty with a diamond-tipped needle placed in the anterior third of the T9 vertebral body. With real-time fluoroscopic guidance, PMMA cement was deliberately directed upward toward the cage at T6–T8. The cement crossed several disk spaces and surrounded the cage anteriorly with some minor filling of the cage.
The second patient had undergone partial corpectomy at L3 with placement of a left paracentral interbody device and dorsal fixation from L2 and L4. Pain recurred approximately 7 months after surgery. MR and CT imaging demonstrated a new compression fracture at L1 and lucencies around the L2–L4 construct. After vertebroplasty was performed at L1, attention was turned to L3. A right parapedicular injection was used to deliver PMMA cement across the midline to the left side anterior to the cage, then a transpedicular injection delivered additional cement to the right side of remaining L3 vertebral body, also anterior to the cage.
Both patients had significant reductions in pain and at 18 months, the constructs demonstrated stability and patients had continued pain relief.22
30.5.4 Adult Spinal Deformity
Adult patients with spinal deformity and long-segment (>5 levels) instrumented fusions are at high risk of proximal junctional kyphosis (PJK), PJFs, and hardware failure (▶Fig. 30.2). This is particularly true in patients with deformity in the sagittal plane. While the specific etiology of these junctional problems is often multifactorial, they are generally due to the stress riser that naturally occurs at the junction of the instrumented and uninstrumented spine.
This is particularly true at the upper instrumented vertebra (UIV) and the vertebra immediately cephalad to the construct (UIV+1). Junctional kyphosis is differentiated into PJK and PJF. PJK is defined as kyphosis of greater than 10 degrees in the segment above a long construct compared with preoperative measurements, while PJF is defined as a structural failure of the spinal column with vertebral body fracture, failure of the posterior ligamentous complex, screw pullout, and vertebral subluxation. Vertebral augmentation is used to prevent both PJK and PJF and has been shown to be effective.23 Hart et al24 revealed a 15% reduction in the incidence of PJF following kyphoplasty cranial to the UIV. Martin et al25 reported that only 5% of patients who underwent vertebral augmentation at UIV and UIV+1 had PJF, much lower than the historic rate without vertebral augmentation. Theologis and Burch12 went further to report that patients with UIV and UIV+1 augmentation had fewer revisions due to fractures (0 vs. 19), better functional outcomes, and significantly less disability. Patients without augmentation were 9.2 times more likely to undergo revision surgery than those with prophylactic UIV and UIV+1 augmentation. Ghobrial et al13 reported a decreased incidence (23.7 vs. 36%) and magnitude (5.65 vs. 9.36 degrees) of PJK in patients with UIV and UIV+1 kyphoplasties. In addition to the early clinical studies examining the effectiveness of vertebral augmentation in the prevention of junctional kyphosis, biomechanical studies have been done as well. Cadaveric analyses of augmentation at UIV and UIV+1 (▶Fig. 30.1) demonstrated fewer PJFs (17%) than those with only a single-level vertebroplasty (that was not at the UIV or UIV+1 level) (67%) or spines without cement (100%)26 (▶Fig. 30.3).
Fig. 30.2 Proximal junctional kyphosis (PJK) (area within black oval) following a lengthy instrumented fusion. (This image is provided courtesy of Dr. Steven M. Theiss, MD.)
30.5.5 Method of Delivery
Cement delivery may be made through standard transpedicular delivery with injection directly into the cancellous bone or via balloon kyphoplasty techniques with augmentation prior to the placement of pedicle screws. This allows the cement to cure or harden around the screws. Pedicle screws placed in osteoporotic vertebrae had higher pullout loads when augmented with the kyphoplasty technique compared to vertebroplasty augmentation (1414 +/− 338 vs. 756 +/− 300 N, respectively; p < 0.001).27 In addition, an unpaired t-test showed that fatigued pedicle screws in osteoporotic vertebrae augmented by kyphoplasty showed higher pullout resistance than those placed in healthy control vertebrae (p = 0.002).27 Both kyphoplasty-type augmentation (p = 0.007) and vertebroplasty augmentation (p = 0.02) increased pullout loads compared to pedicle screws placed in non-augmented osteoporotic vertebrae when tested after fatigue cycling.27
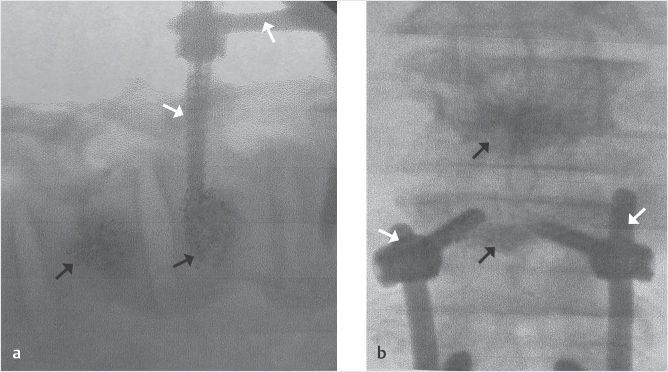
Fig. 30.3 Lateral (a) and anteroposterior (b) fluoroscopic views show vertebral augmentation with polymethyl methacrylate (PMMA) at upper instrumented vertebra (UIV) and UIV+1 (black arrows) in a patient who has previously undergone pedicle screw and rod augmentation (white arrows). (This image is provided courtesy of Fred Parsons, RT.)
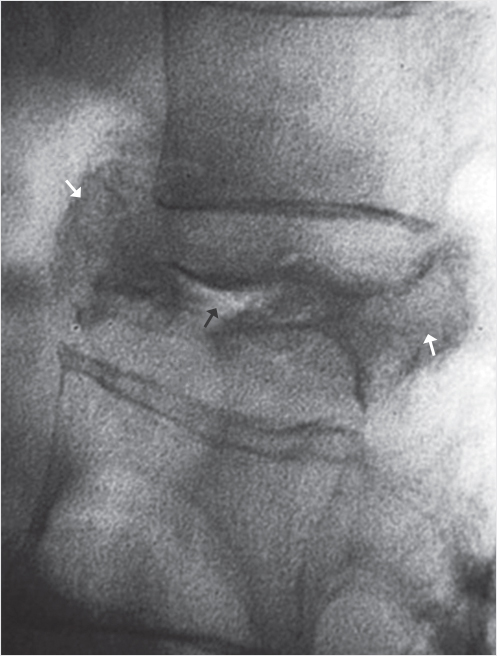
Fig. 30.4 Examples of fenestrated pedicle screw used for polymethyl methacrylate (PMMA) delivery to cancellous bone.29 The screw (black arrow in a) has a central channel (black arrow in b) with fenestrations (black arrowheads) through which PMMA can be injected (white arrowheads in c).
Alternatively, cannulated or fenestrated pedicle screws may be placed and then cement injected through the screws. Cement injected through a cannulated screw is injected into the vertebral body while cement injected into a fenestrated screw surrounds the screw by the PMMA flowing through the fenestrations within the pedicle screw28 (▶Fig. 30.4).
During PMMA delivery through screws with different numbers of side holes (fenestrations), Chen et al30 noted (1) a large amount of PMMA flowed out from the oppositely arranged proximal side holes, whereas almost no PMMA was observed in the distal holes; and (2) the nearer the proximal side hole was to the screw head the greater the risk of cement leakage.30 Published results comparing the conventional needle injection of cement technique around the pedicle screws to delivery of the cement via cannulated screws indicate that although the pullout strength and screw backout torque was significantly higher in the needle injection group (234.1 vs. 187.8 N, 1119.6 vs. 836.7 N mm, respectively), the operation time was shorter and the cement leakage rate was less in the cannulated pedicle screw group (211.4 vs. 296.3 min, 14.05 vs. 26.2%, respectively).31,32
The number, design, and specific layout of the fenestrations of the screw may affect pullout strength.33 Based on the biomechanical performance of various cement-augmented cannulated screw designs, Tolunay et al32 declared in 2015 that the unilateral, sequential, three-radial hole, drilled, cannulated screw was the optimal alternative when considering pullout and torsional strength as criteria. In the same year, Dai et al34 described “a new approach for the application of PMMA augmentation of bone cement-injectable cannulated pedicle screws.” Forty-three consecutive patients with osteoporosis and a degenerative disk disease who had failed at least 6 months of conservative therapy underwent fusion with a novel bone cement-injectable cannulated pedicle screw (CICPS) (Kanghui Medical Devices Co., Ltd., Jiangsu, China). All patients were followed for a mean of 15.7±5.6 months (range: 6–35 months). The Visual Analog Scale and Oswestry Disability Index scores showed a significant reduction in back pain (p = 0.018) and an improvement in lower extremity function (p = 0.025) in patients who underwent lumbar fusion using the novel screw. Intraoperative cement leakage occurred in four patients, but no neurological complications were observed. Radiological observation indicated no loosening or pulling out of the novel screw, and the bone fusion was excellent.
30.5.6 Thoracolumbar Burst Fractures
While cement augmentation of pedicle screw instrumentation can improve the fixation of the screws as described above, other methods of improving fixation in traumatic burst fractures (▶Fig. 30.5 and ▶Fig. 30.6) have been investigated. Norton et al35 performed a biomechanical analysis of treatment for thoracolumbar burst fractures, comparing a four-screw construct (instrumentation immediately above and below the fracture site) to a six-screw construct (pedicle instrumentation of the burst fracture and one level above and one level below). The authors noted a high rate of failure of fixation and early loss of reduction following the four-screw posterior short-segment instrumentation technique. Additional pedicle screws at the level of the fracture (six-screw construct) provided results superior to those utilizing only a four-screw construct. As the additional fixation at the level of the fracture was hypothesized to increase stiffness and reduce the stress on other pedicle screws, a more recent evaluation examined the addition of vertebral augmentation to the four-screw construct compared to the six-screw construct without cement. Clinical outcomes were similar but the traditional six-screw construct was advantageous in terms of shorter operating times, lower blood loss, and lower failure rates.36 However, finite element analysis (FEA) demonstrated that short-segment fixation with two intermediate pedicle screws (the six-screw construct) together with cement at the fractured vertebrae may provide a stiffer construct and less stress on the pedicle screws and rods as compared to other types of short-segment fixation.37 This has yet to be clinically demonstrated. The ability of augmentation of the fractured vertebrae to aid in reduction of traumatic deformity has been investigated. Oner et al38 showed in a cadaveric burst model that balloon-assisted end-plate reduction resulted in predictable reduction of the end plate and kyphosis when combined with vertebral augmentation.38 However, vertebral augmentation combined with posterior instrumentation does not necessarily prevent kyphosis recurrence. Aono et al39 showed that kyphosis recurrence following four-screw constructs can occur even when combined with vertebral augmentation. This is not a result of collapse of the fractured vertebral body, but rather collapse of the adjacent disk, which is injured at the time of the traumatic episode. End-plate reduction and vertebral augmentation though does not affect ultimate disk space collapse.39
Fig. 30.5 Lateral fluoroscopic view shows a burst fracture with the anterior and posterior portions of the vertebral body wider in the anteroposterior dimension than the vertebral body above (white arrows). The image also shows air within the vertebral body (black arrow).
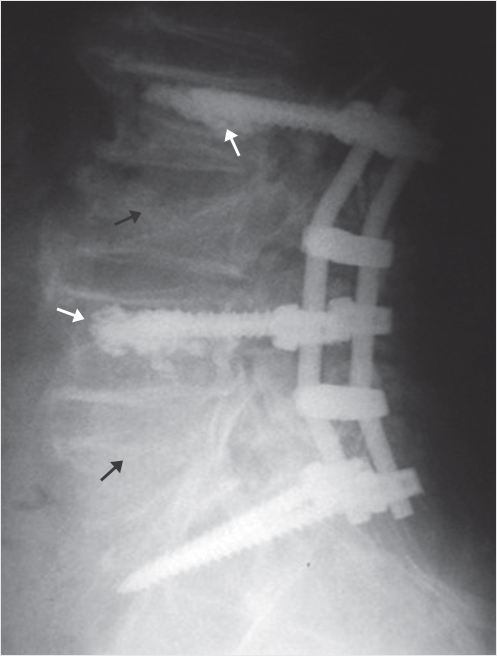
Fig. 30.6 Lateral radiograph shows pedicle screw and rod construct stabilized with polymethyl methacrylate (PMMA) at the level above the L3 fracture and at L4 below the upper fracture (white arrows) and the vertebral compression fractures (VCFs) (black arrows) that are spanned by the construct.
30.5.7 Cement
In vitro data have suggested that augmentation of pedicle screws with PMMA redistributes the pullout forces from the interface of the bone and screw alone to the entire augmented vertebral body and surrounding cortex.11
Cement Composition
Chapter 7 provides a detailed discussion of the various cements and fill materials. PMMA is the most popular bone cement used in vertebral augmentation. The development of PMMA began in 1928 and was first used in orthopaedic surgery in the mid-1950s. It was FDA-approved for treating VCFs in 2004. PMMA is inexpensive, has excellent biocompatibility, and has been associated with minimal long-term complications. However, cardiovascular complications including unpredictable hypotension can occur with its use,28,40 and it may aggravate cardiovascular deterioration in the event of pulmonary cement embolism by stimulating coagulation.41
Other less commonly used bone cements include calcium phosphate (CP), calcium sulfate (CS), calcium triglyceride (CT), and magnesium phosphate (MP). Calcium-based cements typically require 24 hours for curing and thus do not enhance fixation at the time of surgery. They have a lower viscosity and therefore a higher potential for extravasation, although clinical relevance has not been demonstrated.28 In biomechanical analyses, PMMA may have a higher pullout strength and resistance to failure.20,42,43
Cement Volume
While most studies of vertebroplasty describe cement volumes between 2 and 5 mL, many reports note that cement volumes ranging between 2 and 3 mL maximize the initial pullout strength of the screws and that higher volumes may increase the likelihood of cement extravasation.19,44 In a biomechanical analysis comparing pullout strength after standard transpedicular vertebroplasty vs. kyphoplasty, higher volumes of cement were correlated with increased pullout strength.27,33 In 2016, Tan et al33 investigated the optimum injection volume of PMMA to augment a then-novel fenestrated pedicle screw with diameter-tapered perforations. They evaluated how the distribution of cement affected the biomechanical performance of the screw. Study results indicated 1.5 mL of PMMA was a conservative volume for augmentation and that more cement would significantly increase the risk of cement leakage.
Timing of Cement Placement
Based on the in vitro data suggesting that augmentation of pedicle screws with PMMA redistributes the corrective pullout forces from the interface of the bone and screw alone to the entire augmented vertebral body and surrounding cortex, and the theory that screw failure occurs at the bone-cement interface when screws are placed in “soft” cement, it has been suggested that allowing the cement to partially cure prior to screw placement may improve outcomes.11,20 CP cement takes up to 24 hours to harden and therefore does not allow for placement of screws into “hardened” cement, but PMMA, which cures more quickly, may be the best cement for pedicle screw augmentation.28
Regarding the order of screw placement and augmentation, Zapałowicz et al described the ease and benefits of performing vertebral augmentation after the placement of screws with good outcomes.45 Screw placement was followed by extrapedicular vertebroplasty in 22 vertebrae. Radiographically, 73% of the cement was placed centrally and 95% of the vertebral bodies were filled to greater than 70% of their height. There was only a single screw that was not at least partially surrounded by cement. There was a 45% rate of extravasation, but none were symptomatic or clinically relevant.
30.6 Conclusion
Instrumentation has long been used to treat spinal instability. Decades of design modifications have led to improved screw purchase, shorter construct strength, and better fusion outcomes. Vertebral augmentation following instrumentation may further improve outcomes in select patients. Used in combination with pedicle screw fixation, vertebral augmentation offers special promise treating pain and spinal instability in osteoporotic and cancer patients. These patients have elevated risks of hardware failure, collapse of constructs, and junctional fractures.
Outcomes of fusions with vertebral augmentation may vary by etiology, instrumentation, and cement. The cement composition, volume, timing, and method of delivery may all affect success. Biomechanical studies demonstrate improved pullout strength of augmented pedicle screws compared to non-augmented screws in the lumbar and thoracic spine, ilium, and sacrum of osteoporotic patients. Augmentation has been shown to decrease the need for revisions and reduce the risk of PJFs following instrumentation in adults with spinal deformities.
Numerous investigators have correlated outcomes with volumes and methods of cement delivery as well as the biochemical and mechanical properties of available bone cements. PMMA is the most commonly used bone cement and, based on its physical properties, may be the best cement for pedicle screw augmentation. The volume of cement delivered is limited by the risk of leakage. Cement delivery may be made using standard vertebroplasty or balloon kyphoplasty techniques prior to the placement of pedicle screws or via cannulated and/or fenestrated screws. Although there are reports noting the ease and benefit of performing vertebral augmentation after the placement of screws, placement of cement prior to the screws allows the cement to cure or harden around the screws. This may facilitate redistribution of the corrective pullout forces from the interface of the bone and screw alone to the entire augmented vertebral body and surrounding cortex.
Vertebral augmentation is known to reduce pain and disability and improve the quality of life in patients with painful vertebral fractures. In addition to its applications across a wide range of etiologies, it offers promise in select patients when used in combination with instrumented fixation for spinal instability.
References
[1] Roy-Camille R, Saillant G, Berteaux D, Salgado V. Osteosynthesis of thoraco-lumbar spine fractures with metal plates screwed through the vertebral pedicles. Reconstr Surg Traumatol 1976;15(15):2–16
[2] Roy-Camille R, Sailant G, Bisserie M. Surgical treatment of spinal metastatic tumors by posterior plating and laminectomy. In: Proceedings of the 51st Annual Meeting of the American Academy of Orthopaedic Surgeons; 1984
[3] Roy-Camille R, Sailant G, Lapresle P, et al. A secret in spine surgery: the pedicle. In: Proceedings of the 51st Annual Meeting of the American Academy of Orthopaedic Surgeons; 1984
[4] Roy-Camille R, Saillant G, Mazel C. Internal fixation of the lumbar spine with pedicle screw plating. Clin Orthop Relat Res 1986(203):7–17
[5] Kabins M, Weinstein J. The history of vertebral screw and pedicle screw fixation. Iowa Orthop J 1991;11:127–136
[6] Lorenz M, Zindrick M, Schwaegler P, et al. A comparison of single-level fusions with and without hardware. Spine 1991;16(8, Suppl):S455–S458
[7] Thomsen K, Christensen FB, Eiskjaer SP, Hansen ES, Fruensgaard S, Bünger CE. 1997 Volvo Award winner in clinical studies. The effect of pedicle screw instrumentation on functional outcome and fusion rates in posterolateral lumbar spinal fusion: a prospective, randomized clinical study. Spine 1997;22(24):2813–2822
[8] Groff MW. Introduction: guideline update for the performance of fusion procedures for degenerative disease of the lumbar spine. J Neurosurg Spine 2014;21(1):1
[9] Soshi S, Shiba R, Kondo H, Murota K. An experimental study on transpedicular screw fixation in relation to osteoporosis of the lumbar spine. Spine 1991;16(11):1335–1341
[10] Mermelstein LE, McLain RF, Yerby SA. Reinforcement of thoracolumbar burst fractures with calcium phosphate cement: a biomechanical study. Spine 1998;23(6):664–670, discussion 670–671
[11] Sarzier JS, Evans AJ, Cahill DW. Increased pedicle screw pullout strength with vertebroplasty augmentation in osteoporotic spines. J Neurosurg 2002;96(3, Suppl):309–312
[12] Theologis AA, Burch S. Prevention of acute proximal junctional fractures after long thoracolumbar posterior fusions for adult spinal deformity using 2-level cement augmentation at the upper instrumented vertebra and the vertebra 1 level proximal to the upper instrumented vertebra. Spine 2015;40(19): 1516–1526
[13] Ghobrial GM, Eichberg DG, Kolcun JPG, et al. Prophylactic vertebral cement augmentation at the uppermost instrumented vertebra and rostral adjacent vertebra for the prevention of proximal junctional kyphosis and failure following long-segment fusion for adult spinal deformity. Spine J 2017;17(10):1499–1505
[14] Liu D, Wu ZX, Pan XM, et al. Biomechanical comparison of different techniques in primary spinal surgery in osteoporotic cadaveric lumbar vertebrae: expansive pedicle screw versus polymethylmethacrylate-augmented pedicle screw. Arch Orthop Trauma Surg 2011;131(9):1227–1232
[15] Yu BS, Li ZM, Zhou ZY, et al. Biomechanical effects of insertion location and bone cement augmentation on the anchoring strength of iliac screw. Clin Biomech (Bristol, Avon) 2011;26(6):556–561
[16] Yu BS, Zhuang XM, Zheng ZM, Zhang JF, Li ZM, Lu WW. Biomechanical comparison of 4 fixation techniques of sacral pedicle screw in osteoporotic condition. J Spinal Disord Tech 2010;23(6):404–409
[17] Hoppe S, Loosli Y, Baumgartner D, Heini P, Benneker L. Influence of screw augmentation in posterior dynamic and rigid stabilization systems in osteoporotic lumbar vertebrae: a biomechanical cadaveric study. Spine 2014;39(6): E384–E389
[18] Zindrick MR, Wiltse LL, Widell EH, et al. A biomechanical study of intrapeduncular screw fixation in the lumbosacral spine. Clin Orthop Relat Res 1986(203):99–112
[19] Frankel BM, D’Agostino S, Wang C. A biomechanical cadaveric analysis of polymethylmethacrylate-augmented pedicle screw fixation. J Neurosurg Spine 2007;7(1):47–53
[20] Renner SM, Lim TH, Kim WJ, Katolik L, An HS, Andersson GB. Augmentation of pedicle screw fixation strength using an injectable calcium phosphate cement as a function of injection timing and method. Spine 2004;29(11): E212–E216
[21] Kiner DW, Wybo CD, Sterba W, Yeni YN, Bartol SW, Vaidya R. Biomechanical analysis of different techniques in revision spinal instrumentation: larger diameter screws versus cement augmentation. Spine 2008;33(24):2618–2622
[22] Puri AS, Erdem E. Salvage percutaneous vertebral augmentation in failed spinal interbody fusions associated with multiple myeloma. Spine J 2010;10(8): e5–e10
[23] Hyun SJ, Lee BH, Park JH, Kim KJ, Jahng TA, Kim HJ. Proximal junctional kyphosis and proximal junctional failure following adult spinal deformity surgery. Korean J Spine 2017;14(4):126–132
[24] Hart RA, Prendergast MA, Roberts WG, Nesbit GM, Barnwell SL. Proximal junctional acute collapse cranial to multi-level lumbar fusion: a cost analysis of prophylactic vertebral augmentation. Spine J 2008;8(6):875–881
[25] Martin CT, Skolasky RL, Mohamed AS, Kebaish KM. Preliminary results of the effect of prophylactic vertebroplasty on the incidence of proximal junctional complications after posterior spinal fusion to the low thoracic spine. Spine Deform 2013;1(2):132–138
[26] Kebaish KM, Martin CT, O’Brien JR, LaMotta IE, Voros GD, Belkoff SM. Use of vertebroplasty to prevent proximal junctional fractures in adult deformity surgery: a biomechanical cadaveric study. Spine J 2013;13(12):1897–1903
[27] Burval DJ, McLain RF, Milks R, Inceoglu S. Primary pedicle screw augmentation in osteoporotic lumbar vertebrae: biomechanical analysis of pedicle fixation strength. Spine 2007;32(10):1077–1083
[28] Hoppe S, Keel MJ. Pedicle screw augmentation in osteoporotic spine: indications, limitations and technical aspects. Eur J Trauma Emerg Surg 2017;43(1):3–8
[29] Lubansu A, Rynkowski M, Abeloos L, Appelboom G, Dewitte O. Minimally invasive spinal arthrodesis in osteoporotic population using a cannulated and fenestrated augmented screw: technical description and clinical experience. Minim Invasive Surg 2012;2012:507826
[30] Chen LH, Tai CL, Lai PL, et al. Pullout strength for cannulated pedicle screws with bone cement augmentation in severely osteoporotic bone: influences of radial hole and pilot hole tapping. Clin Biomech (Bristol, Avon) 2009;24(8):613–618
[31] Chang MC, Kao HC, Ying SH, Liu CL. Polymethylmethacrylate augmentation of cannulated pedicle screws for fixation in osteoporotic spines and comparison of its clinical results and biomechanical characteristics with the needle injection method. J Spinal Disord Tech 2013;26(6):305–315
[32] Tolunay T, Arslan K, Yaman O, Dalbayrak S, Demir T. Biomechanical performance of various cement-augmented cannulated pedicle screw designs for osteoporotic bones. Spine Deform 2015;3(3):205–210
[33] Tan QC, Wu JW, Peng F, et al. Augmented PMMA distribution: improvement of mechanical property and reduction of leakage rate of a fenestrated pedicle screw with diameter-tapered perforations. J Neurosurg Spine 2016;24(6):971–977
[34] Dai F, Liu Y, Zhang F, et al. Surgical treatment of the osteoporotic spine with bone cement-injectable cannulated pedicle screw fixation: technical description and preliminary application in 43 patients. Clinics (São Paulo) 2015;70(2):114–119
[35] Norton RP, Milne EL, Kaimrajh DN, Eismont FJ, Latta LL, Williams SK. Biomechanical analysis of four-versus six-screw constructs for short-segment pedicle screw and rod instrumentation of unstable thoracolumbar fractures. Spine J 2014;14(8):1734–1739
[36] Liao JC, Fan KF. Posterior short-segment fixation in thoracolumbar unstable burst fractures: transpedicular grafting or six-screw construct? Clin Neurol Neurosurg 2017;153(153):56–63
[37] Liao JC, Chen WP, Wang H. Treatment of thoracolumbar burst fractures by short-segment pedicle screw fixation using a combination of two additional pedicle screws and vertebroplasty at the level of the fracture: a finite element analysis. BMC Musculoskelet Disord 2017;18(1):262
[38] Oner FC, Verlaan JJ, Verbout AJ, Dhert WJ. Cement augmentation techniques in traumatic thoracolumbar spine fractures. Spine 2006;31(11, Suppl):S89–S95, discussion S104
[39] Aono H, Ishii K, Tobimatsu H, et al. Temporary short-segment pedicle screw fixation for thoracolumbar burst fractures: comparative study with or without vertebroplasty. Spine J 2017;17(8):1113–1119
[40] Shridhar P, Chen Y, Khalil R, et al. A review of PMMA bone cement and intra-cardiac embolism. Materials (Basel) 2016;9(10)
[41] Ding T, Yang H, Maltenfort M, Xie R. Silk fibroin added to calcium phosphate cement to prevent severe cardiovascular complications. Med Sci Monit 2010;16(9):HY23–HY26
[42] McLachlin SD, Al Saleh K, Gurr KR, Bailey SI, Bailey CS, Dunning CE. Comparative assessment of sacral screw loosening augmented with PMMA versus a calcium triglyceride bone cement. Spine 2011;36(11):E699–E704
[43] Moore DC, Maitra RS, Farjo LA, Graziano GP, Goldstein SA. Restoration of pedicle screw fixation with an in situ setting calcium phosphate cement. Spine 1997;22(15):1696–1705
[44] Fölsch C, Goost H, Figiel J, Paletta JR, Schultz W, Lakemeier S. Correlation of pull-out strength of cement-augmented pedicle screws with CT-volumetric measurement of cement. Biomed Tech (Berl) 2012;57(6):473–480
[45] Zapałowicz K, Godlewski B, Jekimov R, Grochal M. Augmentation of transpedicular screws by intraoperative vertebroplasty. Neurol Neurochir Pol 2012;46(6):560–568