Major Events in the Evolution of Land Plants
Peter R. Crane and Andrew B. Leslie
OUTLINE
1. Phylogenetic framework
2. Origin and diversification of early land plants
3. Origin and diversification of vascular plants
4. Origin and diversification of seed plants
5. Origin and diversification of angiosperms
6. Innovation in the land plant body
7. Innovation in land plant reproduction
8. Coevolution with animals
9. Patterns of extinction
Although land plants represent merely one branch in the eukaryotic tree of life, they are essential to the energetics and functioning of terrestrial ecosystems. Land plants appear to have arisen from a single colonization of the land surface around 450 million years ago. In the early phases of this colonization, plant innovations centered on the elaboration of a new kind of plant body capable of withstanding the rigors of life on land and exploiting the new opportunities that terrestrial existence provided. Subsequently, this phase of vegetative innovation was followed by successive transformations of the reproductive system, for example, resulting in seeds and flowers, which facilitated increasingly efficient reproduction and dispersal. The unfolding of land plant diversification from a single origin makes it possible to understand all living groups in a relatively simple phylogenetic framework, in which increasingly less inclusive groups are characterized by successive innovations in plant structure and biology.
GLOSSARY
Alternation of Generations. A type of life cycle with multicellular organisms in both the haploid and diploid phases. The diploid phase (sporophyte) produces haploid spores by meiosis that later germinate and develop into the haploid phase (gametophyte), which produces gametes by mitosis. Gamete fusion (fertilization) gives rise to a diploid zygote and marks the transition from the haploid to diploid phase.
Heterospory. The earliest land plants, as well as many living “pteridophytes,” were homosporous, meaning that their sporophytes produced haploid spores of a single size that could each germinate to produce bisexual gametophytes. In contrast, several extinct and extant lineages are heterosporous, meaning they produce two types of spores: larger megaspores, which develop into gametophytes (megagametophytes) that produce only female gametes, and smaller microspores, which develop into gametophytes (microgametophytes) producing only male gametes. Typically, these two types of spores are produced in separate sporangia (megasporangia and microsporangia, respectively).
Seed. The seed can be considered an extreme form of heterospory in which a single megaspore develops within a megasporangium that never opens to release the spore. In seed plants, this megasporangium is further enveloped by a protective covering called the integument, and the whole structure is termed an ovule. The ovule becomes a seed on fertilization, and at maturity it contains an embryo, an accompanying food source, and a protective seed coat.
Vascular Tissue. Vascular tissues are specialized cells that transport water and nutrients through the plant body. The xylem, or water-conducting tissue, is composed principally of cells called tracheids (or similar but modified cells called fibers or vessels) that are dead at maturity and have walls that are typically thickened by the deposition of the complex biopolymer lignin. The phloem, which conducts sugar and nutrients through the plant, contains living transport cells called sieve cells as well as other cell types.
1. PHYLOGENETIC FRAMEWORK
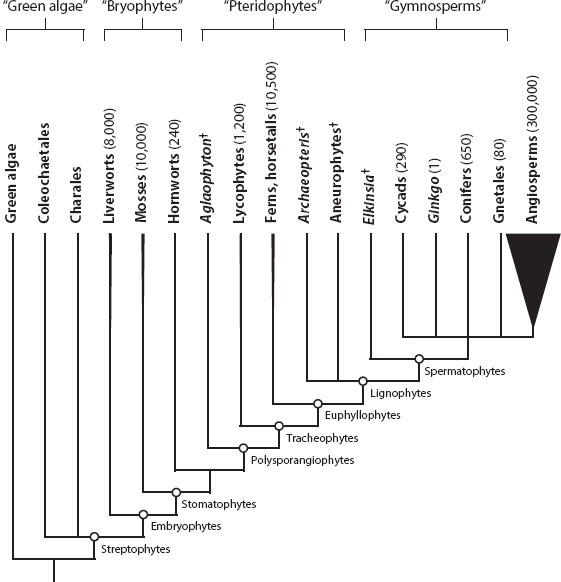
A phylogenetic framework in which to understand the 11 major groups of living land plants was originally developed from structural data and has been confirmed by analyses based on molecular sequences (figure 1). Plants are a monophyletic subgroup of eukaryotes defined by the presence of the photosynthetic pigments chlorophylls a and b, the storage of starch in plastids (chloroplasts), and several other features. In addition to land plants, this definition includes freshwater and marine organisms often grouped together as “green algae” and contrasts with treatments in which plants are defined more narrowly as synonymous with land plants. Molecular and structural data, including characteristics of cell division, support the placement of certain freshwater “charophycean green algae” (Coleochaetae, Chara) as the closest relatives of land plants; these groups together form a clade called the streptophytes.
Figure 1. Phylogenetic framework for the evolution of land plants, including the 11 major living land plant groups and several extinct groups (indicated by a dagger) discussed in the text. The general topology is based on results from analyses of morphological data, which are broadly supported by results from molecular data sets. Major clades are indicated by a hollow circle; important clades discussed in the text include embryophytes (land plants), tracheophytes (vascular plants), and spermatophytes (seed plants). Paraphyletic groups, such as “bryophytes,” are indicated in quotes. The relationships among the “bryophyte” groups have been controversial, and this topology reflects the current hypothesis based on analyses of several large molecular data sets. Likewise, relationships among the five living groups of seed plants remain controversial, with different molecular data sets generating different topologies, especially in regard to the position of the Gnetales. Here these relationships have been left unresolved. The approximate number of species in each group of extant land plants is given after the group name. The thickness of the line for each living plant group is scaled to reflect its proportion of total land plant species diversity (groups with fewer than 1000 species are depicted with the same line thickness). Note the overwhelming diversity of angiosperms (flowering plants) compared with all other groups; approximately 90% of all living terrestrial plant species are angiosperms.
The land plant clade, or embryophytes, is composed of several major monophyletic groups that are widely used in discussions of plant evolution (figure 1), including vascular plants (tracheophytes), seed plants (spermatophytes), and flowering plants (angiosperms). There are also corresponding paraphyletic groups (see chapter II.1) at each level. For example, “green algae” are those plants that are not land plants, “bryophytes” are those land plants that are not vascular plants, “pteridophytes” are those vascular plants that are not seed plants, and “gymnosperms” are those seed plants that are not flowering plants. Analyses of molecular data suggest that living “gymnosperms” (conifers, cycads, Ginkgo, Gnetales) may make up a monophyletic group, although when extinct seed plants are included, “gymnosperms” are clearly paraphyletic. Within flowering plants, phylogenetic analyses support monocots (such as grasses and palm trees) as a monophyletic group, but “dicots” are now known to be paraphyletic. Two major monophyletic groups, eudicots and eumagnoliids, together account for almost all species previously treated as dicots.
2. ORIGIN AND DIVERSIFICATION OF EARLY LAND PLANTS
Land plants are distinguished from “green algae” by a suite of reproductive and vegetative innovations, many of which are important in overcoming shortages of water. In land plants, the egg is formed and fertilized in a specialized flask-like structure called the archegonium within which the young embryo begins its development. Land plants also have desiccation-resistant spore walls impregnated with the complex biopolymer sporopollenin; this enables their reproductive progagules to survive drying and disperse among suitable environments. The land plant body is covered in a waxy cuticle that prevents water loss, and all land plant groups except liverworts also have stomata, pores in the cuticle that are used to regulate the exchange of gases and water vapor with the atmosphere.
Spore-like microfossils that may have been produced by plants or other eukaryotes are known from as early as the middle Cambrian (ca. 510 Ma), although their exact affinities are unclear. Spores that were most likely produced by embryophytes appear around the Middle Ordovician (ca. 465 Ma), with forms broadly similar to those of some living “bryophytes.” Beginning around the same time, and continuing through much of the Silurian (ca. 443–416 Ma), the fossil record preserves a variety of enigmatic organic sheets and tubes suggestive of land plant cuticle and simple water-conducting tissues. Such structures provide further evidence that the diversification of early land plants was under way, even though our knowledge of the plants involved is extremely fragmentary (see chapter II.10).
Macroscopic evidence of terrestrial plants is scarce until the middle to late Silurian (ca. 420 Ma), when small branching axes bearing multiple sporangia appear in the fossil record. These fossils represent the first-known appearance of a major clade, the polysporangiophytes, which exhibit an important innovation in the vegetative plant body. While living “bryophyte” groups (hornworts, liverworts, mosses) produce a morphologically diverse array of sporophytes, they are all small, unbranched structures bearing a single sporangium at their tip. In contrast, the branched sporophytes of polysporangiophytes can produce multiple sporangia from a single embryo. Unlike the situation in “bryophytes,” the larger sporophytes of at least some early polysporangiophytes were also probably nutritionally independent of their corresponding gametophyte. Polysporangiophyte fossils become increasingly common in the latest part of the Silurian (ca. 416 Ma) and into the Early Devonian, where a glimpse into early polysporangiate diversity is provided by the classic earliest Devonian Rhynie Chert locality in Scotland (ca. 413 Ma). This assemblage preserves an early terrestrial ecosystem in exquisite detail, including arthropods, fungi, polysporangiophytes, and early representatives of a polysporangiophyte subgroup, the vascular plants, which were to become the most important constituent of later terrestrial ecosystems.
3. ORIGIN AND DIVERSIFICATION OF VASCULAR PLANTS
Living vascular plants are distinguished from hornworts, liverworts, and mosses by their specialized vascular tissues (see glossary) and their larger and more elaborate sporophyte. Rhynie Chert polysporangiophytes exhibit a mixture of features that appear to reflect various stages in the evolution of these tissues and structures. The gametophyte and sporophyte of at least some Rhynie Chert plants were similar in size and morphological complexity, which is unlike the life cycle of any living terrestrial plant and may reflect an intermediate stage between the small sporophytes of “bryophytes” and the earliest polysporangiophytes (some Cooksonia species) and the more elaborate sporophytes of true vascular plants. Some Rhynie Chert polysporangiophytes (e.g., Aglaophyton, Horneophyton) contain water-conducting tissues that are similar to the unspecialized conducting cells found in some living mosses, while others (Rhynia) have water-conducting cells with pronounced internal thickenings but lack the extensive deposition of the resistant biopolymer lignin characteristic of modern tracheids. Paleobotanists once grouped plants like Aglaophyton, Horneophyton, and Rhynia together as Rhyniophytes, but their structural features suggest they are most likely of diverse relationships: some are likely on the stem lineage leading to true vascular plants (tracheophytes), while others may represent early branches within the tracheophytes.
Asteroxylon, a vascular plant from the Rhynie Chert with modern-type tracheids, appears to be related to living lycophytes, an early branch of vascular plant evolution distinguished by their kidney-shaped sporangia and characteristic pattern of vascular tissue development. The Early and Middle Devonian fossil record (ca. 416–385 Ma) shows that lycophytes diversified quickly into two main groups: zosterophylls, which were diverse during the earliest phases of vascular plant evolution but became extinct by the end of the Devonian, and lycopods, which diversified through the Devonian and include about 1200 living species that occur in a wide range of modern biomes. One extinct lycopod subgroup included large trees (greater than 40 m tall in some cases) that dominated the well-known Pennsylvanian tropical coal swamps of Eurasia and North America. While most of these arborescent lycopods were extinct by the end of the Permian (ca. 251 Ma), some members of this lineage survived into the Mesozoic. The only living representative of this group is the genus Isoetes, a small rosette plant of wet places.
Lycophytes shared the Early and Middle Devonian landscape with “trimerophytes,” a poorly understood but important assemblage of plants with naked, variously branched photosynthetic axes bearing elongated terminal sporangia. In basic vegetative construction, “trimerophytes” were similar to Rhynie Chert plants such as Aglaophyton, Horneophyton, and Rhynia, but they often had more complex branching patterns and include forms such as the Early Devonian plant Pertica, which had a well-defined central axis and lateral branches. While the morphology and anatomy of some “trimerophytes” is quite well characterized, their relationships to each other and to living groups are poorly understood; nevertheless, these plants are thought to represent the early evolution of the second major lineage of vascular plants (the euphyllophytes; lycophytes being the other) that ultimately gave rise to several groups of ferns, living and extinct horsetails (relatives of living Equisetum), and seed plants.
The exact pattern of relationships among these groups is controversial; molecular data suggest that living ferns, horsetails, and Psilotales (a group of simple, fernlike plants) form a monophyletic group to the exclusion of seed plants while analyses of morphological data incorporating fossil taxa do not always support this relationship. Whatever the precise pattern of relationships, indisputable horsetails, with their characteristic jointed stems and whorled leaves, are first known from the Late Devonian (ca. 385–359 Ma), and from then on this lineage is a ubiquitous feature of the plant fossil record. As in lycopod evolution, some groups (called calamites) attained the stature of large trees during the Mississippian and Pennsylvanian; however, despite the long evolutionary history of this group, their morphological and ecological diversity has been relatively limited; both extinct and extant forms tend to favor wet environments.
Ferns have a complicated evolutionary history characterized by several successive waves of diversification. There are diverse possible fern relatives in the Middle and Late Devonian and a number of probable early fern groups in the Mississippian, although none of these plants can be readily assigned to living groups. However, ferns closely related to living Marattiales were prominent components of terrestrial ecosystems in the Pennsylvanian and formed a major component of many coal-swamp environments.
Most groups of modern ferns (termed leptosporangiate ferns or Filicales) belong to a clade with a characteristic pattern of sporangial development in which a single initial cell gives rise to the sporangium. A number of Carboniferous fern groups have been included in this lineage based on their sporangial morphology and stem anatomy, although their exact phylogenetic relationships are unclear. Early diverging extant Filicales (e.g., the living family Osmundaceae) first appear in the late Permian (ca. 251–244 Ma), and plant assemblages during the Mesozoic often include representatives of other early diverging extant filicalean groups such as Dicksoniaceae, Dipteridaceae, Gleicheniaceae, Matoniaceae, Marsileaceae, and Schizaeaceae. While these groups contain relatively few species today, they were important components of Jurassic and Cretaceous plant communities. Over the Late Cretaceous and Cenozoic, certain subgroups of filicalean ferns (the polypod ferns) have undergone a further major radiation that accounts for most extant fern diversity.
One additional group of Middle and Late Devonian free-sporing plants developed an important structural innovation that had major consequences for the subsequent history of life on land. These “progymnosperms” (e.g., Aneurophyton, Archaeopteris) had a layer of actively dividing cells in their axes capable of producing a cylinder of secondary xylem (wood) to the inside and cylinder of secondary phloem to the outside. The presence of this feature, called a bifacial cambium, defines the lignophyte clade that also includes all extinct and living seed plants. Although many groups of vascular plants have developed some form of secondary growth, a bifacial cambium allowed lignophytes to increase the girth of their axes (through the production of wood) while maintaining connections for the transport of nutrients throughout the plant (through the secondary phloem). Large, long-lived sporophytes that could potentially grow for an indeterminate amount of time were one result of this innovation, and by the Late Devonian (ca. 380 Ma), “progymnosperms” such as Archaeopteris had sporophytes similar in basic structure to modern seed plant trees such as living conifers.
4. ORIGIN AND DIVERSIFICATION OF SEED PLANTS
While early lignophytes like Archaeopteris exhibited vegetative features similar to those of seed plants, their reproductive biology was fundamentally the same as that of “bryophytes,” lycopods, ferns, and horsetails. All these groups reproduce by spores dispersed through the air which then germinate to produce a free-living haploid gametophyte (see glossary). Fertilization occurs when motile male gametes released by gametophytes swim through films of water in the environment and fertilize eggs in the archegonia on the same or nearby gametophytes. Seed plants, however, have a very different kind of reproductive biology in which female gametophytes develop on the parent plant inside structures called ovules (see glossary). Seed plant reproductive biology therefore includes a unique process called pollination where small male gametophytes (contained within pollen grains) are transported to the ovules by wind, water, or animals. After pollen germination, the male gametophyte grows and ultimately produces gametes that will fertilize an egg cell in the female gametophyte. After fertilization, the embryo begins to grow inside the ovule, which is now termed a seed. This radically new kind of reproductive biology, seen in five living groups (conifers, cycads, Ginkgo, Gnetales, and angiosperms), created new opportunities for maternal investment in offspring, gave sporophytes greater control over the reproductive process by minimizing the influence of the outside environment, and allowed seed plants to colonize a much greater variety of terrestrial habitats by reducing dependence on external films of water for fertilization and enclosing embryos within a protective coat. These advantages have likely contributed to the ecological dominance of seed plants in most terrestrial ecosystems since the Permian.
The earliest fossils that can be recognized unambiguously as seed plants (e.g., Archaeosperma, Elkinsia, Moresnettia) date from the latest Devonian (ca. 365 Ma) and are generally grouped together within a potentially paraphyletic group, the “hydraspermans,” characterized by particular type of pollination biology. The diversity of hydrasperman and other early seed plant groups increases through the Mississippian and Pennsylvanian, although many of these plants had not yet acquired the full suite of features seen in living seed plant groups; for example, their pollen grains germinated in a manner similar to that of “pteridophyte” spores. Seed plants with fully modern seed plant reproduction, including probable early conifers and cycads as well as several now-extinct groups, do not appear until the Pennsylvanian and Permian.
Seed plants continued their taxonomic and ecological radiation through the Mesozoic. In addition to familiar groups such as cycads, Ginkgo, and several families of conifers, Mesozoic fossil assemblages contain a wide diversity of extinct groups that were ecologically important at different times and in different parts of the world (e.g., Bennettitales, Caytoniales, Corystospermales). The phylogenetic relationships of many of these extinct seed plant groups are uncertain; for example, analyses based on morphological data usually resolve the groups Bennettitales, Erdtmanithecales, and Gnetales as closely related to each other, and in turn closely related to flowering plants. However, molecular data from the limited set of extant seed plants suggest instead that Gnetales are more closely related to conifers, and that cycads and Ginkgo also belong to this group to the exclusion of angiosperms.
5. ORIGIN AND DIVERSIFICATION OF ANGIOSPERMS
Nearly all seed plant diversity, and therefore nearly all terrestrial plant diversity as a whole, comprises flowering seed plants, or angiosperms (figure 1). While the phylogenetic position, and hence the origin, of angiosperms relative to other seed plant groups remains uncertain, knowledge of phylogenetic patterns within the group has been clarified substantially by advances in molecular phylogenetics. Angiosperms are distinguished from other seed plants by a suite of innovations in both vegetative and reproductive biology that favor efficiency and speed in functionality, growth, and development. Angiosperm leaves are characterized by multiple, reticulate vein orders, and the stems of most species contain specialized xylem elements called vessels. Both these features allow angiosperms to rapidly and efficiently move water through the plant body, although they have both apparently originated convergently in a few other groups of living and extinct seed plants (for example, in living Gnetum and extinct gigantopterids).
It is in reproductive biology, however, that the differences between angiosperms and other seed plants are most pronounced. Angiosperm ovules and seeds are borne inside a closed structure called the carpel so that unlike other groups of seed plants, angiosperm pollen grains land and germinate away from the ovules on a specialized receptive tissue called a stigma. Angiosperm ovules also typically have two integuments (see glossary) rather than one as in most other seed plants. The female gametophyte of angiosperms is very simple and presumably highly reduced (typically seven cells with eight nuclei) and fertilization is also unique: in most species one male gamete fuses with the egg, while another fuses with a diploid nucleus in the female gametophyte. The triploid nucleus resulting from this second fertilization event develops to form the nutritive tissues of the seed, the endosperm. Through this process of double fertilization the development of the nutritive tissue is linked directly to successful formation of the zygote.
Unequivocal angiosperm flowers, with a carpel or ovary surrounded by characteristic pollen-producing organs (stamens) and a perianth (petals or similar structures), are first recorded in the fossil record around the middle of the Early Cretaceous (ca. 125 Ma). Unambiguous angiosperm pollen grains with complex pollen walls structurally similar to those of living taxa occur somewhat earlier (ca. 135 Ma). In the Late Cretaceous and continuing through the Cenozoic, the diversity of angiosperm species and their ecological importance has increased explosively. Angiosperms are by far the most diverse group of terrestrial plants, and they ultimately gave rise to forms as diverse as grasses, palms, water lilies, orchids, oaks, and sunflowers. Angiosperms are the autotrophic foundation of modern terrestrial ecosystems; through their intricate and complex interactions with microbes, fungi, animal herbivores, pollinators, and seed dispersers, their diversification has been of fundamental importance in the origin of terrestrial biodiversity as a whole. The diversification of angiosperms may have even made possible the occupation of new kinds of habitats and the origin of new kinds of terrestrial biomes. For example, the increased water-cycling rates made possible by angiosperm leaves may have contributed to the development of extensive rain forests in the tropics.
6. INNOVATION IN THE LAND PLANT BODY
In very broad terms, the history of innovation in land plants can be divided into two phases: a period of intense vegetative innovation from about the Late Ordovician to the Late Devonian (450–360 Ma) and a period of reproductive innovation that began around the Late Devonian and still continues. The first phase is marked by a suite of adaptations relating to the acquisition, retention, and transport of water and other fluids and also includes a strong trend toward increasing the size and stature of the sporophyte. This elaboration of the sporophyte was most likely driven by the advantages of large size for increased reproductive output and by competition for light as the most favorable ecological sites on land became more crowded. One manifestation of this trend can be seen in the rooting structures of Silurian and Devonian plants, which become increasingly elaborate and more efficient at gathering water and nutrients from the soil compared with those of the earliest land plants and modern “bryophytes,” in which delicate root hairs (called rhizoids) collect water but do not deeply penetrate the soil. Increasing root penetration may also have significantly increased terrestrial weathering rates, potentially leading to a drawdown of atmospheric CO2 and global cooling in the Late Devonian and Carboniferous.
These increasingly complex rooting structures served to support increasingly large and complex aerial plant bodies. Much morphological and anatomical evolution between the late Silurian and Late Devonian consisted of various groups developing larger, more ramified, and more specialized photosynthetic axes, resulting in plants such as Middle and Late Devonian Rhacophyton and Pseudosporochnus (generally considered relatives of early ferns) with complex, three-dimensional arrays of photosynthetic branches that in some ways mimic large compound leaves. From the Late Devonian through the Pennsylvanian, multiple lineages (horsetails, ferns, “progymnosperms,” seed plants) independently developed laminar leaves with specialized photosynthetic tissue between the leaf veins. Many of the same lineages also developed large arborescent forms on which these new photosynthetic organs were arrayed. While there were certainly important innovations in vegetative structure through the late Paleozoic, Mesozoic, and Cenozoic (for example, the origin of vessels and high vein density in angiosperms, or the reorganization of the plant body seen in monocotyledons), the basic vegetative structure of modern terrestrial plants was already established by the Late Devonian.
7. INNOVATION IN LAND PLANT REPRODUCTION
Following the basic innovation of the branching sporophyte by the late Silurian, the reproductive biology of terrestrial plants appears to have changed little over the Devonian, even though their vegetative structure became increasingly complex. While several lineages developed heterospory (see glossary and further reading), reproduction remained fundamentally based on the simple release of wind- or water-dispersed spores; however, beginning in the Late Devonian and continuing through the Mississippian and Pennsylvanian, there was a diversification of reproductive morphology associated with the emergence, radiation, and rise to ecological dominance of seed plants. In seed plants, reproduction is more complicated than the dispersal of spores; reproductive success depends on pollen output, the number of ovules, the efficiency with which pollen can reach the ovules, and the efficacy of subsequent fertilization, embryo development, and seed dispersal. The result was a diverse array of morphologies linked to the dispersal of gametes (accomplished through pollination) and the dispersal of embryos (contained in the seeds).
Reproductive innovation increased still further after the Pennsylvanian, and the evolutionary history of seed plants (especially angiosperms) can be thought of as a nearly limitless exploration of ways to more efficiently invest reproductive resources, move pollen, disperse seeds, and ensure the survival of offspring. Some aspects of this evolutionary history reflect basic advances in the mechanics of terrestrial reproduction. For example, the pollen grains of early seed plants and some living gymnosperms such as cycads and Ginkgo produce motile sperm that swim through liquid inside ovules, similar to the way motile male gametes of ferns swim through films of water on the forest floor; however, in groups such as angiosperms, conifers, and Gnetales, the motile phase is lost entirely and male gametes are delivered directly to eggs by tubes that develop from the pollen grains. Resource allocation in seeds has also become generally more efficient through time; early groups of seed plants such as medullosan seed ferns from the Pennsylvanian developed nearly fully provisioned seeds (technically ovules) before fertilization, and perhaps before pollination, had occurred. In contrast, most flowering plants make almost no investment in the growth of ovules or seeds until after fertilization.
Other aspects of seed plant reproductive evolution reflect responses to constantly changing ecological interactions with pollinators and seed dispersers. For example, fleshy cone tissues that attract vertebrate seed dispersers, usually birds, have evolved independently in several groups of living conifers. Similarly, “flowerlike” reproductive structures occur in insect-pollinated living “gymnosperms” (e.g., Welwitschia in the Gnetales) and in extinct “gymnosperms” that were most likely insect pollinated (most Bennettitales) as well as in flowering plants. In angiosperms, structural innovations such as the bisexual flower and the carpel function in conjunction with complex pollination and seed dispersal interactions involving insects, birds, and mammals, and such interactions have long been thought to be centrally important in generating the enormous diversity in the group.
8. COEVOLUTION WITH ANIMALS
Plant evolution has unfolded in an ecological theater that was constantly being rebuilt by the changing positions of oceans and continents, as well as by major fluctuations in climate. In addition, there have been major changes in the reciprocal interactions between animals, plants, and microbes as all three groups have evolved. For example, the earliest terrestrial ecosystems appear to have been very different from those we would recognize today; fossil evidence suggests plants were largely free from animal herbivory, and the earliest Silurian and Devonian insects seem to have been primarily detritivores or carnivores. Similarly, the earliest terrestrial vertebrates in the Devonian, Mississippian, and Pennsylvanian show no obvious adaptation in dentition or body size for the direct consumption of plants (see chapters II.17 and II.18). This implies that the flow of energy in Devonian ecosystems was very different from that in modern ecosystems; fungi that actively decomposed plants and insects that fed on this decomposed plant matter probably occupied a central place in a food web with few direct links between animals and plants. Direct insect herbivory, however, was well established by the Pennsylvanian, and the intensity and complexity of plant-herbivore interactions has continued to increase over time. The first unequivocal vertebrate herbivores are recorded in the early Permian and have likewise increased in diversity and ecological importance through time, leading ultimately to the extremely large dinosaur herbivores of the Jurassic and Cretaceous and the large mammalian herbivores of the Cenozoic that replaced them (see chapter II.18). The evolution of large grazing mammals together with changing climates during the later Cenozoic may also have helped create the open habitats that have been exploited very effectively, and relatively recently, by derived groups of herbaceous angiosperms (e.g., grasses, Asteraceae).
Early indications of potential interactions between plants and pollinators come from pollen found in coprolites and gut contents of Mesozoic insects. At the same time, the aggregation of ovules and pollen-producing organs into a single flowerlike structure, as seen, for example, in extinct Bennettitales, may have created opportunities for insects to deposit and receive pollen during the same visit; however, direct evidence of plant-pollinator interactions is sparse until the rise of angiosperms, and it is only within this group that animal pollination has been exploited to its fullest potential and highest degree of specialization. While Early and mid-Cretaceous flowers are not highly specialized compared with those of many modern taxa, most were clearly pollinated by insects (probably beetles and flies). Beginning in the Late Cretaceous, and increasing through the Cenozoic, a dramatic radiation of floral morphology has been occurring as increasingly intricate interactions have developed between angiosperms and a huge range of insect pollinators such as bees, beetles, flies, and butterflies as well as birds, bats, rodents, and even primates.
The history of coevolution in relation to seed dispersal broadly parallels that of pollination. Evidence has been found of diffuse interactions between animal dispersers and many Mesozoic plants; for example, the seeds of fossil cycads, Ginkgo, and Caytonia were surrounded by fleshy tissues that may have been important in attracting animal dispersers, as they are today in many living “gymnosperms.” Among angiosperms, indications of relatively generalized interactions with animal dispersers first appear during the Early Cretaceous. Many Cretaceous angiosperms produced small fleshy fruits that suggest some kind of animal dispersal; however, in the early Cenozoic, there is an abrupt increase in both fruit and seed size that appears to be linked with the availability of bird and mammal dispersers as well as with a change from relatively open plant communities to more dense vegetation and closed forest canopies. Such environments may have favored larger seeds with more stored nutrients for the establishment of seedlings. In general, the great variety of fruit and seed morphology seen among living angiosperms is linked to an equally great variety of interactions with animal seed dispersers.
9. PATTERNS OF EXTINCTION
The fossil record provides ample evidence of extinction in the history of plants on land, just as it does in the history of terrestrial animals, but successive episodes of mass extinction seem to have had much less influence on plant evolution. A few significant perturbations in the history of plant life do coincide with major extinction events in the animal record; for example, the apparent loss of the ecologically important plant group glossopterids at the Permian-Triassic boundary, and the significant loss of species diversity in North America at the Cretaceous-Paleogene boundary. These episodes often appear linked to regional climatic changes, however, rather than to fundamental shifts in the composition of global vegetation. There are also equally large perturbations that do not coincide with major extinctions in the animal world, for example, the loss of coal swamps and their characteristic plants toward the end of the Paleozoic, which seems to have been caused by increasingly arid climates. It also appears that the great diversity in some groups of plants, for example, angiosperms and certain groups of filicalean ferns, may reflect low extinction rates as well as high speciation rates. In the broadest sense, the major changes in terrestrial vegetation through time appear to reflect a more gradual pattern of displacement by competition among plant groups rather than repeated resetting of the evolutionary clock by mass extinction. Large-scale evolutionary patterns among plants seem to predominantly reflect successive biological innovations that resulted in increased vegetative and reproductive efficiency against a background of changing ecological conditions.
FURTHER READING
Algeo, T. J., and S. E. Scheckler. 1998. Terrestrial-marine teleconnections in the Devonian: Links between the evolution of land plants, weathering processes, and marine anoxic events. Philosophical Transactions of the Royal Society B 353: 113–130. Discusses increases in terrestrial weathering rates that may have accompanied the innovation of deeply rooted plants in the Devonian and provides an example of the broad effects of plant structural evolution on terrestrial ecosystems.
Bateman, R. M., and W. A. DiMichele. 1994. Heterospory: The most iterative key innovation in the evolutionary history of the plant kingdom. Biological Reviews 69: 345–417. Summarizes the evolution of heterospory in different vascular plant lineages and offers potential explanations for its frequent occurrence.
Boyce, C. K. 2008. How green was Cooksonia? The importance of size in understanding the early evolution of physiology in the vascular plant lineage. Paleobiology 34: 179–194. Offers a new interpretation of the physiology of some of the earliest polysporangiophytes and discusses the evolution of the sporophyte in early terrestrial plants.
Boyce, C. K., T. J. Brodribb, T. S. Feild, and M. A. Zwieniecki. 2009. Angiosperm leaf vein evolution was physiologically and environmentally transformative. Proceedings of the Royal Society B 276: 1771–1776. This study suggests that angiosperm vegetative innovations, in particular their high leaf vein density relative to other groups of vascular plants, may have altered terrestrial ecosystems by increasing the rate of water cycling and therefore contributing to the formation of extensive tropical rain forest biomes.
Friis, E. M., K. R. Pedersen, and P. R. Crane. 2011. Early Flowers and Angiosperm Evolution. Cambridge: Cambridge University Press. A summary and synthesis of the known information about early angiosperm ecology, biology, and floral morphology based on Cretaceous fossils and their living relatives.
Kenrick, P., and P. R. Crane. 1997. The Origin and Early Diversification of Land Plants: A Cladistic Study. Washington, DC: Smithsonian Institution Press. A summary, synthesis, and analysis of the morphology, anatomy, and systematics of early vascular plants and their living relatives.
Mathews, S. 2009. Phylogenetic relationships among seed plants: Persistent questions and the limits of molecular data. American Journal of Botany 96: 228–236. Summarizes the various hypotheses of relationships among living seed plant groups based on molecular and morphological data sets and discusses some potential reasons for the variety of results.
Pryer, K. M., E. Schuettpelz, P. G. Wolf, H. Schneider, A. R. Smith, and R. Cranfil. 2004. Phylogeny and evolution of ferns (monilophytes) with a focus on the early leptosporangiate divergences. American Journal of Botany 91: 1582–1598. This study proposes a general hypothesis for relationships among living “pteridophytes” based on a large molecular data set.
Shaw, A. J., P. Szövényi, and B. Shaw. 2011. Bryophyte diversity and evolution: Windows into the early evolution of land plants. American Journal of Botany 98: 352–369. Summarizes the current state of research on the diversity and systematics of bryophytes, especially in relation to the early evolution of land plants.